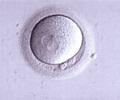
The first baby to be created from an egg matured in a laboratory, frozen, thawed and then fertilised, has been born in Canada, researchers told a medical conference on Monday.
The baby girl was born to a woman diagnosed with advanced ovarian disease, and three other women in the 20-person trial group are pregnant by the same technique, researchers said.Doctors already collect and store eggs from women who face cancer treatment that could cause sterility.
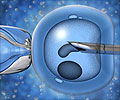
The eggs -- harvested after stimulating the ovaries with hormones -- are fertilised in-vitro with their partner's sperm, then frozen. After the cancer therapy, the eggs are then thawed and implanted.
The Canadian team said the prototype technique took this fertility preservation a major step further. The eggs were recovered from unstimulated ovaries and in addition were fertilised after they had been frozen and then thawed, and not before.
"Until now, it was not known whether oocytes (human eggs) collected from unstimulated ovaries, matured in vitro and then vitrified could survive thawing, be fertilised successfully and result in a viable pregnancy after embryo transfer," said Hananel Holzer, who led the team from the McGill Reproductive Center in Montreal.
"We have demonstrated for the first time that it is possible to do this and, so far, we have achieved four successful pregnancies, one of which has resulted in a live birth. The other three pregnancies are ongoing."
Advertisement
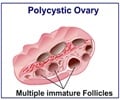
The trial group comprised 20 women, whose average age was about 30, who had been diagnosed by ultrasound to have polycystic ovarian syndrome (PCOS), a fertility-threatening hormonal disorder that affects around 10 percent of all women of child-bearing years.
Advertisement
They were then matured in the laboratory for 24-48 hours, and were frozen for several months and then thawed. Of these, 148 oocytes survived the thawing process and were fertilised by a technique called intra-cytoplasmic sperm injection.
Sixty-four embryos were then transferred to the women, who received multiple embryos in order to boost the chance of a successful pregnancy.
Holzer said his team had progressively improved the success rate, noting that three of the pregnancies had been achieved in the past five patients.
But he cautioned against giving rise to false hopes. The research is still in its preliminary stages and had not yet been proven in cancer patients, the biggest potential beneficiaries.
Women who face chemotherapy or radiotherapy for cancer may not have time to undergo ovarian stimulation so that their oocytes can be harvested and stored.
Their only other option for preserving their fertility is the still-experimental option of having ovarian tissue removed before they undergo cancer therapy.
The tissue is frozen and then transplanted back at a later date, although some experts worry that this may also reimplant cancer cells.
For women who are infertile with PCOS, taking hormones to stimulate egg production can lead to potentially fatal overstimulation of the ovaries.
Thus, obtaining immature eggs from unstimulated ovaries, and then coaxing them towards a pregnancy, is a major step forward for patients in this group.
Separately, ESHRE reported that scientists in Israel had been able to obtain and freeze eggs from the ovarian tissue of girls as young as five years old.
Childhood cancers usually result in cure rates of between 70 and 90 percent, but sometimes this comes at the cost of inflicting sterility when the child reaches adult life.
Obstetrician Ariel Revel of the Hadassah University Hospital, Jerusalem, found eggs in the ovarian buds of pre-pubescent girls aged between five and 10 years.
The eggs were gently sucked out using a needle, were matured in a lab dish and then frozen, for potential use in the future.
Source-AFP
LIN/M