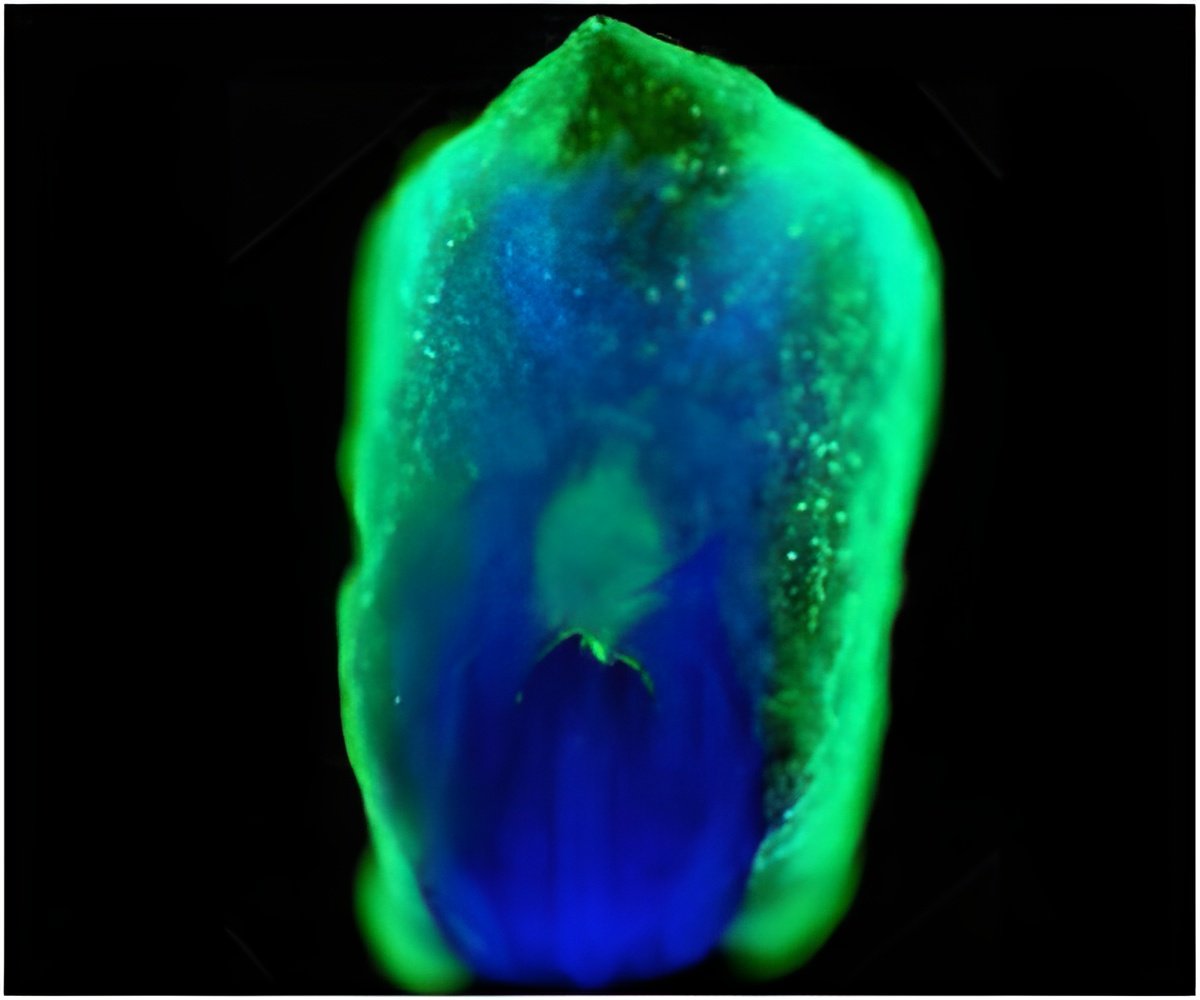
Investigators from the Semmelweis University, the National Institute of Oncology, and the National Koranyi Institute of Pulmonology, Budapest, Hungary, and the Medical University of Vienna, Vienna, Austria, used electron and confocal microscopy to examine tumor tissue in mice in which malignant tumor cells had been introduced. They proposed a novel mechanism for the development of tissue pillars (the most characteristic feature of intussusceptive angiogenesis, in which a vessel folds into itself to form two vessels). Moreover, they demonstrated a significant increase in pillar formation after treatment with the angiogenesis inhibitor vatalanib. Their observations support the notion that inhibition of just a single tumor vascularization mechanism can trigger alternative ones.
Prior to this study, the mechanism of pillar formation had not been fully understood. Investigation revealed a progression of events that generates a connection between the processes of endothelial bridging and intussusceptive angiogenesis resulting in rapid pillar formation from pre-existing building blocks. To describe this mechanism of pillar formation the group coined the term "inverse sprouting."
"It is well established now that tumors can obtain sufficient blood supply from alternative vascularization mechanisms (such as intussusceptive angiogenesis) to grow without capillary sprouting (known as the key mode of new vessel formation in cancer). Therefore, antiangiogenic therapies should be tailored depending on the angiogenic phenotype in each single tumor, and the targeting of non-sprouting angiogenic mechanisms in cancer seems to be a rational strategy. Our study provides new understanding of cancer-induced intussusceptive angiogenesis and may serve as a basis for the development of novel drugs targeting this type of blood vessel formation."
Source-Eurekalert