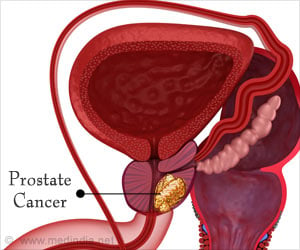
A new prostate cancer "homing device" invented by Purdue University researchers can improve detection, and allow for the first targeted treatment of the disease.
The researchers have revealed that they have synthesized a molecule that finds and penetrates prostate cancer cells, and created imaging agents and therapeutic drugs that can link to the molecule and be carried with it as cargo.Philip Low, the Ralph C. Corley Distinguished Professor of Biochemistry who led the team, said that a targeted treatment could be much more effective in treating cancer and would greatly reduce the harmful side effects associated with current treatments.
"Currently none of the drugs available to treat prostate cancer are targeted, which means they go everywhere in the body as opposed to only the tumour, and so are quite toxic for the patient," said Low, who is a member of the Purdue Cancer Center.
"By being able to target only the cancer cells, we could eliminate toxic side effects of treatments. In addition, the ability to target only the cancer cells can greatly improve imaging of the cancer to diagnose the disease, determine if it has spread or is responding to treatment," Low added.
The Purdue team say that the molecule they have created attaches to prostate-specific membrane antigen (PSMA), a protein that is found on the membrane of more than 90 percent of all prostate cancers.
Low points out that it is also found on the blood vessels of most solid tumours, and may provide a way to cut off the tumour blood supply.
Advertisement
The researcher says that there also is potential for the targeting molecule to be used to attack the vasculature of solid tumours of other types of cancers.
Advertisement
"The molecule acts like a homing device for prostate cancer. PSMA, which is found only on prostate cancer cells and tumor blood vessels, acts as the homing signal that the molecule targets. The molecule and its cargo go only to cancerous tissue, leaving healthy tissue unharmed," says Sumith Kularatne, a graduate student in Purdue's chemistry department and first author of both papers who compared the targeting molecule to a homing device.
He has revealed that the molecule is designed with a specific shape that fits with the protein like a key to a lock. The molecule and its cargo are then carried inside the cell with the protein as it goes through its normal cycle.
A radioimaging application used for body scans is expected to enter clinical trials this fall, and an optical imaging application used to measure prostate cancer cells in blood samples is already in clinical trials.
The findings of the researchers have been described in two research articles published in the journal Molecular Pharmaceutics.
Source-ANI
TAN





![Prostate Specific Antigen [PSA] Prostate Specific Antigen [PSA]](https://www.medindia.net/images/common/patientinfo/120_100/prostate-specific-antigen.jpg)