Thus far the vaccine has been used to treat women with cervical, vulvar, and vaginal cancer, and associated lesions caused by HPV types 6, 11, 16, and 18.
"Treatment for anal cancer is challenging; the use of Gardasil as a method of prevention is important as it may result in fewer diagnoses and the subsequent surgery, radiation or chemotherapy that individuals need to endure," said Karen Midthun, director of the FDA Center for Biologics Evaluation and Research.
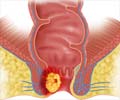
Although anal cancer is rare in the general population, its occurence is increasing especially among gay men, officials noted.
Human papillomavirus (HPV) is associated with about 90% of the estimated 5,300 cases of anal cancer diagnosed in the United States each year, the FDA said, citing statistics from the American Cancer Society. More women are diagnosed than men.
But Gardasil won't prevent development of anal precancerous lesions already present at the time of vaccination, the FDA said.
Advertisement
Source-Medindia