Duncan Wardrop, associate professor of chemistry at University of Illinois at Chicago and corresponding author of the new study, collaborated with UIC virologist Lijun Rong, who created a screening system that uses a chimeric HIV-Ebola virus bearing the protein coat of the Ebola virus. The chimera looks like Ebola but isn't life-threatening for scientists to work with.
After screening more than 230 candidate compounds, Wardrop and Rong found two molecules that inhibited cell entry, but only one that demonstrated specificity for the Ebola virus - plus a bonus.
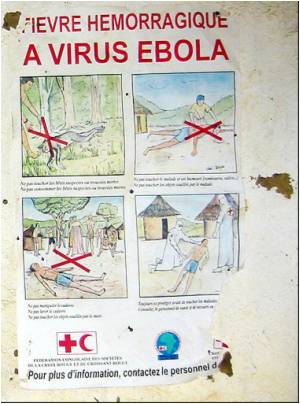
"We found that our lead compound also inhibits Marburg," Wardrop said, referring to a related virus that, along with Ebola, is one of the deadliest pathogens known.
"That was a nice surprise. There's growing evidence the two viruses have the same cell-entry mechanism, and our observations appear to point to this conclusion."
In an effort to find even more potent anti-Ebola agents, Wardrop and graduate student Maria Yermolina synthesized a series of derivatives of the lead molecule-a member of a family of compounds called isoxazoles-and found several that displayed increased activity against Ebola infection.
Advertisement
Source-ANI