GlaxoSmithKline on Thursday announced that FDA has granted a standard 10-month review of its experimental human papillomavirus vaccine Cervarix, Reuters reports. A GSK spokesperson on Thursday said FDA declined to grant priority review, which could have been completed in six months.
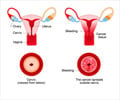
Cervarix and Merck's HPV vaccine Gardasil have been shown to be 100% effective in preventing infection with HPV strains 16 and 18, which together cause about 70% of cervical cancer cases. Researchers in a 2006 study published in the online edition of the Lancet also found that Cervarix prevented infection with HPV strains 31 and 45, which together with strains 16 and 18 cause more than 80% of cervical cancer cases. GSK in January announced that it would begin a trial to determine whether Cervarix provides better protection against cervical cancer than Gardasil.The Australian Therapeutic Goods Administration last week granted the first major market license for Cervarix, approving the vaccine for girls and women ages 10 to 45. According to Reuters, the European Medicines Agency could recommend the vaccine next month. FDA will most likely take action on GSK's application in January 2008, according to analysts at Evolution Securities.
Source-Kaiser Family Foundation
LIN/M