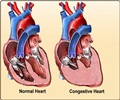
This is only the second such catheter system to be approved in the United States and while treating atrial fibrillation through catheter ablation is still in its infancy, leading medical experts predict that the market will see a big growth in the near future.
“The physician community likes cryo. They consider it safe, and so from that perspective I think Medtronic has a good device on the market and will be able to take market share. I think it's a big deal”, the analyst for Noble Financial Capital Markets, Jan Wald said.
Source-Medindia