New biotechnologies that allow scientists to quickly and accurately distinguish species based on a simple DNA analysis are being creatively deployed for the first time in the war against a major global disease.
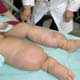
The University of Ghana, supported by the Philadelphia-based JRS Biodiversity Foundation, is pioneering the use of DNA "barcodes" to map menacing mosquito species in West Africa that spread lymphatic filariasis (LF), commonly known as elephantiasis. Using a short DNA sequence from a particular genome region, scientists can obtain a species' 'barcode' identity. Barcodes are needed because closely-related species, with different capabilities to transmit LF, are otherwise hard to distinguish.The ability to precisely identify mosquito species in this way is a promising advance in the battle against LF, an often disfiguring disease that today threatens 1 billion people across roughly 80 countries. Over 120 million people have the parasitic infection and more than 40 million have been permanently disabled or disfigured.
The research is identifying species spreading the worm larvae that clog the human lymph system, often causing grotesque swelling. By revealing the menace species' habitat and range, it also aids understanding of environmental factors that influence their breeding and abundance.
"The scientific breakthrough of DNA barcoding, which grew explosively from a single Canadian research paper in 2003, is shedding new light on LF – a horrific and entirely preventable health scourge in developing countries," says principal investigator Prof. Daniel Boakye of the University of Ghana.
"Beyond the immediate battle against this disease in West Africa, the value to human health of these important new tools will grow as the range and habitats of specific mosquito species shift due to climate change."
LF is a leading cause of permanent and long-term disability worldwide and results from a microscopic, thread-like worm spread between humans through a mosquito's bite. Living within a human's blood, the worm larvae grow into adults which mate and produce other larvae, called microfilariae.
Advertisement
Poor sanitation and rapid growth in tropical and subtropical areas has created more places for mosquitoes to breed and thus to more LF infection.
Advertisement
The drug reduces the density of worm larvae in humans. This LF elimination strategy relies on a belief that the region's main LF vector, the Anopheles mosquito, is incapable of transmitting low-density worm larvae.
But the Anopheles family is highly diverse and contains hundreds of species. And the new molecular studies reveal that not all Anopheles species are created equal. Some can transmit the disease despite the drugs' thinning of the worm larvae.
The research is pointing out places infested with the menace species and, therefore, where the drug strategy needs to be supplemented with insecticides to successfully eliminate LF.
Prof. Boakye also notes that blanket vector control using insecticides can have serious impact on non-target organisms, leading to biodiversity loss. The additional information and insights into specific mosquito species allows for those species and areas to be targeted, reducing the level of spraying and its effect on other organisms.
"The expertise to create databases is in short supply in Africa; the JRS Foundation is assisting science in very meaningful ways," he says.
Source-Eurekalert
LIN