Pharma majors Abbott and Enanta Pharmaceuticals have announced the launching of clinical trials of ABT-450, an oral protease inhibitor for the treatment of chronic HCV. Protease inhibitors can keep a virus from making copies of itself.
"Hepatitis C is a serious global health concern, with 170 million people currently infected by six different HCV genotypes," said John M. Leonard, senior vice president, Global Pharmaceutical Research and Development, Abbott. "As a global leader in the development of antiviral therapies and diagnostics, Abbott is bringing its decades of antiviral experience, particularly with protease inhibitors, to this collaboration and to the fight against HCV.""ABT-450 demonstrated favorable potency in vitro across various HCV genotypes and highly resistant strains," said Jay R. Luly, Ph.D., president and CEO of Enanta Pharmaceuticals. "We look forward to working with Abbott to advance ABT-450, and to our building a pipeline of HCV protease inhibitors that addresses this widespread disease."
The Phase 1, double-blind, placebo-controlled study for ABT-450 announced today is a single, ascending oral dose trial in healthy volunteers.
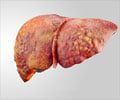
Hepatitis C is a viral disease that is transmitted through contaminated blood. Approximately 25% get rid of the infection while the rest develop a chronic form of the disease. If left unattended, it can progress to liver cirrhosis and liver failure, eventually leading to liver cancer.
Liver disease associated with HCV infection is growing rapidly, and current therapies only provide sustained benefit in about half of patients with the genotype 1 form of the virus. Specifically targeted antiviral therapies for HCV, such as NS3/4a protease inhibitors, may have the potential to increase the proportion of patients in whom the virus can be eradicated.
Enanta Pharmaceuticals is a research and development company and is developing novel protease, polymerase and cyclophilin-based inhibitors targeted against the Hepatitis C virus (HCV), according to a press release. It is a privately held company headquartered in Watertown, Massachusetts.
Abbott is working to advance the treatment of Hepatitis C (HCV) through a multifaceted discovery and development program that leverages the company's deep experience in antiviral medicines. Compounds in various states of development include protease inhibitors (ABT-450) and polymerase inhibitors (ABT-333 and ABT-072.
Advertisement
Source-Medindia
GPL/S