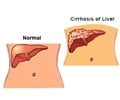
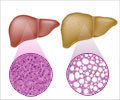
New findings with regard to the renin angiotensin system (RAS) have forced researchers to have a rethink on the mechanism behind liver cirrhosis.
While the angiotensin converting enzyme (ACE)-Ang 2-AT1 receptor arm contributes to liver tissue injury and fibrosis and the maintenance of basal vascular tonus in non-compensated cirrhosis, the activation of the ACE2-Ang-(1-7)-Mas receptor arm exerts anti-fibrotic actions and probably has also a role in arterial vasodilation in liver cirrhosis.In a previous study published last year in WJG by the same research group, it was shown that chronic treatment with propranolol in cirrhotic patients was characterized by marked changes in the precursors of the RAS cascade (renin and Ang 1 with repercussions on the 2 main RAS components, Ang 2 and Ang-(1-7), in the splanchnic and peripheral circulation. Therefore, the circulating profile of RAS components at different stages of liver cirrhosis and their role in hemodynamic changes of cirrhosis remained unclear.
A research article to be published in May 28, 2009 in the World Journal of Gastroenterology addressed this issue. The research team led by Prof. Santos from the Federal University of Minas Gerais measured the circulating levels of angiotensins in patients at different stages of liver cirrhosis by radioimmunoassay, and further evaluated the influence of RAS components on hemodynamic changes during liver transplantation.
The study showed that the progression of liver dysfunction is characterized by marked changes in circulating Ang-(1-7) and Ang 2 levels. In the initial stages, there were a predominance of Ang-(1-7) rather than Ang 2. On the other hand, advanced stages of cirrhosis showed an activation of the peripheral and splanchnic RAS, and a deviation toward the formation of Ang-(1-7) in the splanchnic circulation. Furthermore, there was a positive correlation between the Ang-(1-7)/Ang 2 ratio and cardiac output and a negative correlation between this ratio and systemic vascular resistance, indicating that the final functional effects of the RAS may reflect a balance between these 2 opposing peptides.
The relationship between Ang-(1-7) and Ang 2 may play a role in hemodynamic changes of human cirrhosis. The peptide Ang-(1-7) probably predominates in the peripheral circulation in the initial stages of human cirrhosis and in the splanchnic circulation in the advanced stages, both contributing to a reduction in vascular resistance and consequently to hyperdynamic circulation. In the peripheral circulation of patients with advanced cirrhosis, when compared to the splanchnic circulation, Ang 2 probably leads to extra-splanchnic vasoconstriction. Although further studies with a larger number of patients should address the precise role of RAS in human cirrhosis, this research opens the possibility that future therapies interfering with the RAS in both the systemic and splanchnic circulation should lead to more success in the management of the hemodynamic changes in human cirrhosis.
Advertisement
RAS