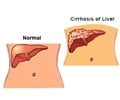
Union Minister for Health and Family Welfare Dr. Anbumani Ramadoss on Tuesday sought the support of India's proposal for observing 2nd October every year as 'World No Alcohol Day'.
Dr. Ramadoss also called for an institution of a framework convention on Alcohol Control on the lines of framework convention on Tobacco Control.Kicking off another round of cooperation between India, Brazil and South Africa (IBSA), Health Ministers of the three countries agreed to collaborate on wide range of health related issues.
The issue of definition of counterfeit medical products was highlighted by India. Dr. Ramadoss urged for a United IBSA Forum "to engage in order to achieve clarity on any new definition of counterfeit drugs that is sought to be introduced".
Pointing to the differences in the laws of various countries, Dr. Ramadoss said "our understanding is that such drugs which are recognized as generics in some counties cannot be considered to be counterfeit, even in those countries where they may not have market authorization. And certainly, the transhipment of such drugs cannot be controlled".
Commenting on registration of drugs and medicine issues, Dr. Ramadoss suggested that joint discussions could be initiated to build mutual confidence by way of standardization and harmonization of the internal approval processes. This will help in reaching consensus on the norms prevailing in the three countries.
Dr. Ramadoss said that in addition to the selected areas of traditional medicine, registration of drugs and medicine strengthening public health laboratories, research and development cooperation can be initiated between ICMR (India), MRC (South Africa) and FIOCRUZ (Brazil) under the aegis of global strategy and plan of action on public health innovations and intellectual property.
Advertisement
SPH