Information about Midostaurin
Generic Name : MidostaurinUp-to-date prescription details regarding Midostaurin are provided here.
Pronunciation : MYE-doe-STAW-rin
Learn the correct pronunciation of the Midostaurin, understand it's uses, recommended dosages, its indications, how to take, when to take, when not to take, side effects, special precautions, warnings and its and its storage instructions.You will also find a list of the medication's International and Indian brand or trade names, as well as its pricing information. For verification of the information presented on this page or for additional clarifications, it's advisable to consult your doctor.
Therapeutic Classification : Chemotherapy
Trade Names/Brand Names of Midostaurin
India :
Rydapt
Overview of Midostaurin
• Midostaurin is an anticancer drug, approved by FDA for the treatment of acute myeloid leukemia (AML) in adult patients.Why is Midostaurin Prescribed? (Indications)
Midostaurin is a kinase inhibitor prescribed as a combination therapy with cytarabine and daunorubicin for treating adult patients with acute myeloid leukemia who are detected as FLT3 mutation-positive by an FDA approved test.It is also used for treating aggressive systemic mastocytosis (ASM), systemic mastocytosis with associated hematological neoplasm (SM-AHN) or mast cell leukemia (MCL).
When should Midostaurin not be taken? (Contraindications)
Midostaurin should not be used in patients with:• Allergy or known hypersensitivity to midostaurin
• Lung-related toxicity with the drug
• Pregnancy and breastfeeding
What is the dosage of Midostaurin?
Acute myeloid leukemia:• The prescribed adult dose is 50 mg orally taken twice daily on day 8 to 21 of each cycle of induction with daunorubicin and cytarabine, and days 8 to 21 days of each cycle of consolidation with a high dose of cytarabine in patients who show FLT3 mutation positivity detected by an FDA approved test.
Treatment of ASM, SM-AHN, MCL:
• The recommended adult dose is 100 mg orally taken twice daily and the treatment should be continued until the disease progression or occurrence of toxicity.
How should Midostaurin be taken?
• Midostaurin comes in a capsule form to be taken by mouth along with food. It should be taken at the same time each day.• The capsules should be swallowed as a whole with sufficient quantity of water.
• Do not crush or open the capsule.
• If vomiting occurs after taking midostaurin, do not take another dose and continue with the next scheduled dose.
What are the warnings and precautions for Midostaurin?
• Patients treated for systemic mastocytosis should be monitored for toxicity for the first 4 weeks followed by every other week for the next 8 weeks and monthly once for the remaining duration of the treatment.• Antiemetic drugs should be used before taking midostaurin mainly to reduce the risk of nausea and vomiting.
• Monitor patients for the signs and symptoms of any lung-related complications or infections of the respiratory tract.
• Treatment of midostaurin should be discontinued in case the blood counts remain low for more than 21 days.
• Midostaurin should not be used as a monotherapy in treating patients with acute myeloid leukemia.
• Men with female partners and women of childbearing age should use effective contraceptive measures during midostaurin therapy and should continue using them for at least 4 months after the last dose of midostaurin to avoid harm to the fetus.
What are the side effects of Midostaurin?
Common: Nausea, vomiting, neutropenia (low white blood cell counts), edema (fluid accumulation), fatigue, feverGastrointestinal: Inflammation of the mucous membranes, piles, raised pancreatic enzymes, abdominal discomfort or pain, constipation, bleeding
Cardiovascular: Prolonged QT interval (an abnormality in the electrical activity of the heart), fluctuations in blood pressure, heart attack, pericardial effusion (fluid accumulation around the heart), clot formation in a blood vessel
Central nervous system: Headache, dizziness, vertigo, insomnia, difficulty in attention, tremor
Blood: Low blood counts
Respiratory: Nasopharyngitis, sinusitis, bleeding from the nose, difficulty in breathing, lung inflammation, pleural effusion (fluid accumulation around the lung), cough, pain in the oropharynx, upper respiratory tract infection, lung toxicity
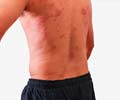
Skin: Skin lesions in the form of petechiae, maculopapular rash, erythema multiforme, and erysipelas increased sweating, dry skin
Others: Swelling of eyelids, herpes virus infection, increased levels of creatinine, joint pain, raised levels of uric acid in blood, muscle pain, high blood glucose levels, device-related infections, increase weight, high blood calcium levels, fever
What are the other precautions for Midostaurin?
• Pregnancy test has to be performed within seven days starting the midostaurin treatment and the drug should be initiated only if the pregnancy test is negative.• Midostaurin may impair fertility in both men and women of childbearing age. It is advised to consider the option of using a sperm or egg bank to store sperms or eggs before starting treatment.
• The use of midostaurin capsules in pediatric patients is not recommended as the safety and effectiveness of the drug has not established in this group.
What are the Drug Interactions of Midostaurin?
• Drugs such as clarithromycin, ketoconazole, diltiazem, idelalisib and antiviral drugs such as ritonavir, saquinavir, tipranavir and nelfinavir increase the concentration of midostaurin when taken together results in toxicity.• Midostaurin concentrations decrease when taken with drugs such as carbamazepine, phenytoin, rifampin, mitotane, and enzalutamide leading to reduced effectiveness of midostaurin therapy.
• Grapefruit juice results in toxicity of midostaurin while St.John’s Wort reduces the midostaurin concentrations when taken concomitantly.
What are the storage conditions for Midostaurin?
• Store midostaurin capsules at a controlled room temperature between 20°C and 25°C.• Keep the capsules in the original container.
• Protect from excess light and moisture.