The team have been awarded Biomedical Catalyst funding from the Medical Research Council (MRC) to take the drug into a clinical trial in multiple myeloma patients, scheduled to begin in late 2015.
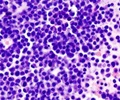
Multiple myeloma is an incurable cancer of the bone marrow, which accounts for nearly two per cent of all cancer deaths.
Professor Guido Franzoso, from the Department of Medicine at Imperial College London, who led the research, said: "Lab studies suggest that DTP3 could have therapeutic benefit for patients with multiple myeloma and potentially several other types of cancer, but we will need to confirm this in our clinical trials, the first of which will start next year."
The new drug was developed by studying the mechanisms that enable cancer cells to outlive their normal lifespan and carry on multiplying. In the 1990s, a protein called nuclear factor kappa B (NF-kB), which plays an important role in inflammation, and the immune and stress response systems, was discovered to be overactive in many types of cancer, and responsible for switching off the normal cellular mechanisms that naturally lead to cell death. This enables the cancer cells to survive.
The pharmaceutical industry and scientists around the world have invested heavily in research into NF-kB inhibitors, but such compounds have not been successfully developed as therapies because they also block the many important processes controlled by NF-kB in healthy cells, causing serious toxic side effects.
Advertisement
By studying cells from multiple myeloma patients, they identified a protein complex, named GADD45β/MKK7, that appeared to play a critical role in allowing the cancer cells to survive.
Advertisement
"We had known for many years that NF-kB is very important for cancer cells, but because it is also needed by healthy cells, we did not know how to block it specifically. The discovery that blocking the GADD45β/MKK7 segment of the NF-kB pathway with our DTP3 peptide therapeutic selectively kills myeloma cells could offer a completely new approach to treating patients with certain cancers, such as multiple myeloma," Professor Franzoso said.
A spinout company, Kesios Therapeutics, was formed to commercialize DTP3 and other drug candidates based on Professor Franzoso's research, with support from Imperial Innovations, a technology commercialization company focused on developing the most promising UK academic research.
"The significant progress made by Professor Franzoso in multiple myeloma is one of the many cancers we believe his signal transduction research could be applied to. To help develop this ground-breaking research further, Imperial Innovations created the spin out Kesios Therapeutics," explained Dayle Hogg from the Healthcare Ventures team at Imperial Innovations.
Source-Eurekalert