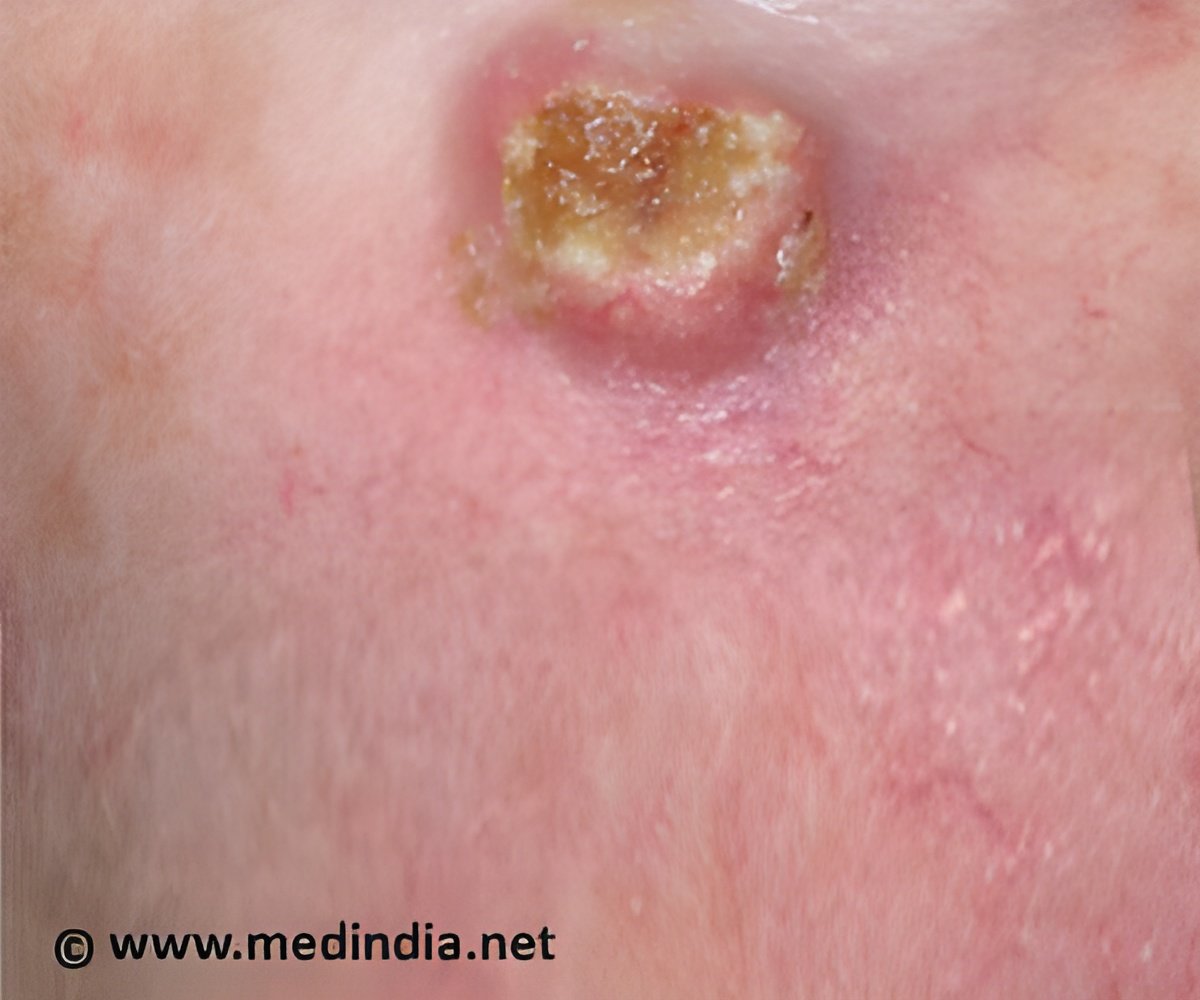
Currently, there is no cure for CTCL aside from a bone marrow transplant.
However, the new study shows that the topical gel can eliminate the cancerous cells.
"The results of the trial suggest that resiquimod is safely and effectively absorbed into the skin, and beyond diminishing treated lesions, also enhances the immune response, leading to healing of even untreated lesions," said first author and principal investigator Alain Rook, professor at Perelman School of Medicine at University of Pennsylvania, Philadelphia, US.
"To our knowledge, this is the first topical therapy that can clear untreated lesions and lead to complete remission in some patients," Rook noted.
The findings offer hope to patients who have not responded to other modalities, including certain types of topical chemotherapy and phototherapy.
Advertisement
Patients applied specified doses to some portions of the affected skin for 16 weeks.
Advertisement
By the final evaluation, treated lesions were significantly improved in 75 percent of patients, and 30 percent saw full resolution in all treated lesions.
Unlike other treatments, resiquimod also improved untreated lesions, resulting in more than 50 percent improvement for more than 90 percent of patients.
Two participants, one of whom had been living with CTCL for more than 15 years without response to treatment, saw full eradication of the disease.
The study was published online in the journal Blood.
Source-IANS















