"This is a really exciting milestone for this research," said John McCarrey, director of the San Antonio Cellular Therapeutics Institute. "This is the first time that anybody has been able to show the concept works in a primate model, and that is an important step in moving the research forward to clinical trials."
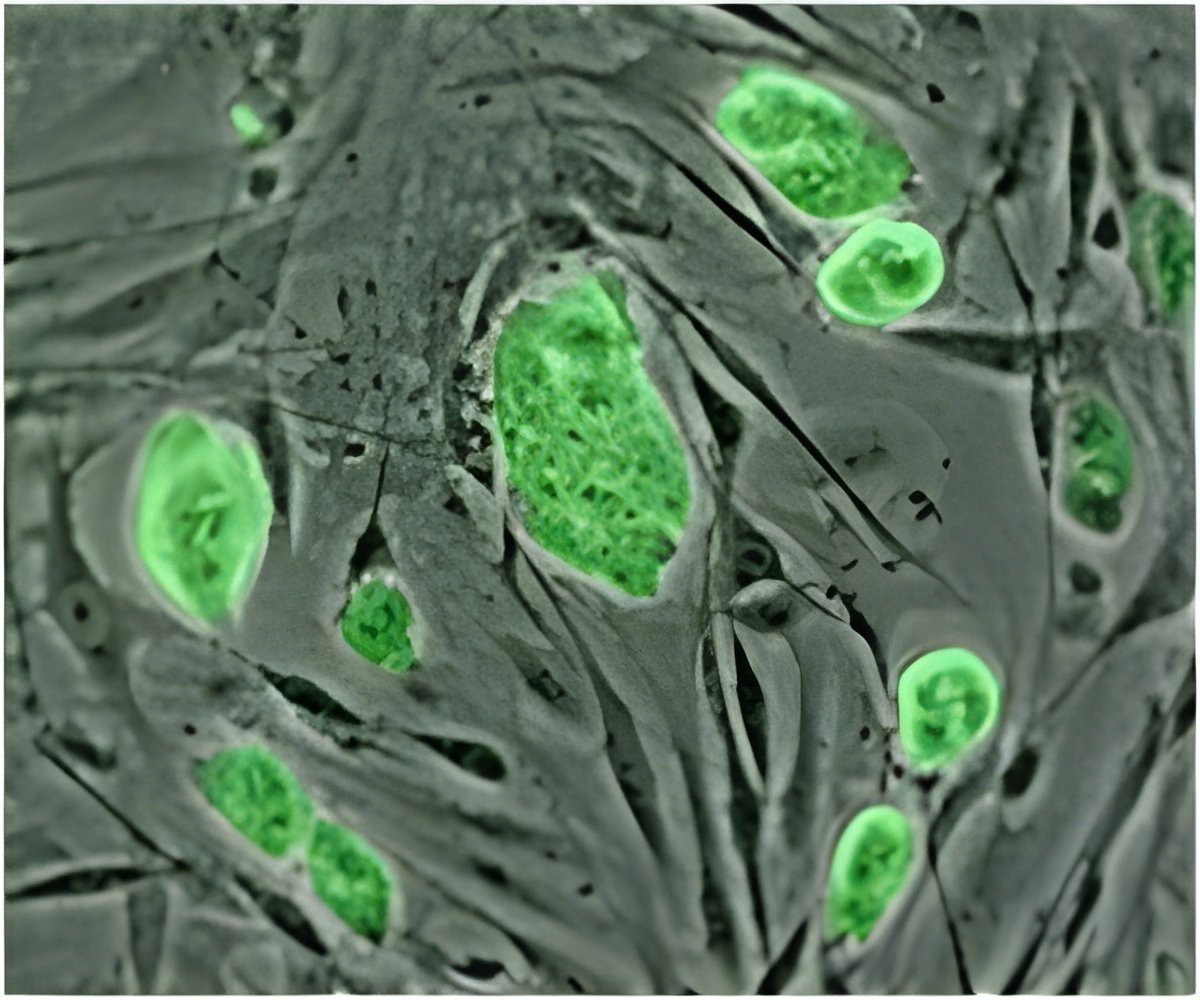
While men facing cancer treatments, which could cause infertility, are able to store their own sperm for future use in the fertility clinic, this is not an option for boys before puberty who are not yet making sperm. But, all prepubertal boys have spermatogonial stem cells (SSCs) in their testes, which could be used for transplantation.
The concept of using spermatogonial stem cells to restore fertility was first introduced in the mid-1990s by University of Pennsylvania scholar Ralph L. Brinster. Since that time, scholars have been working to demonstrate the concept is viable.
But more work is required.
The research must overcome a number of hurdles before it can become a common clinical practice.
Advertisement
But it's hard for researchers to know when clinical trials will begin since the removal and storage of spermatogonial stem cells is currently a rare practice worldwide.
Advertisement
Hermann joined the UTSA College of Sciences' faculty in summer 2011, following a post-doctoral fellowship at MWRI alongside Associate Professor Kyle Orwig. At UTSA, he is continuing to focus his research on basic and translational studies of spermatogonial stem cells to preserve fertility in boys treated for cancer and related diseases.
"For a long time, oncologists have been unable to address the long-term consequences of life-saving chemotherapy and radiation treatments such as infertility," said Hermann. "That is now beginning to change as laboratory research such as this study provides new experimental options for patients facing infertility after cancer."
Source-Eurekalert