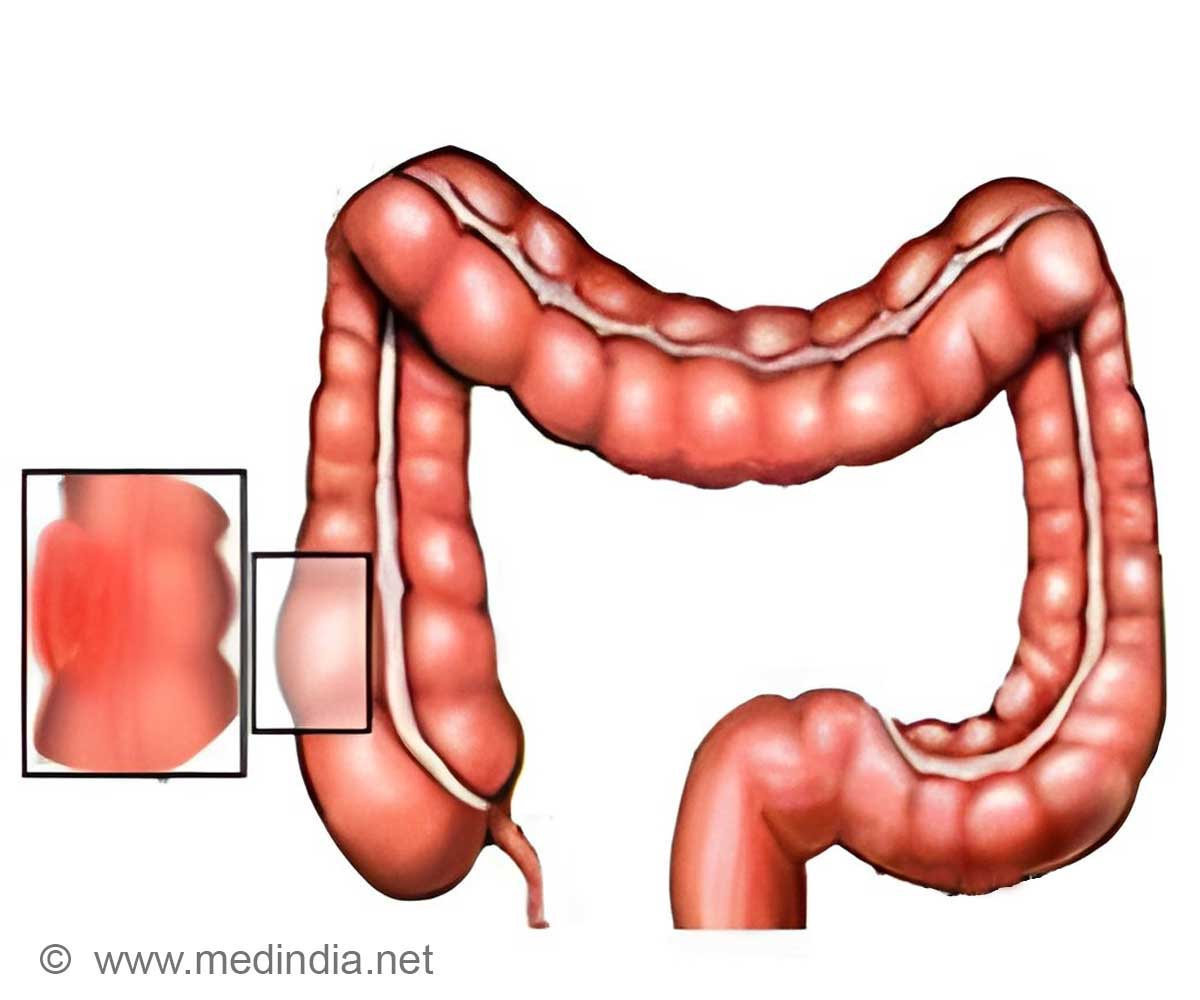
Sanofi revealed that the US Food and Drug Administration has approved its metastatic colorectal cancer drug Zaltrap which has been developed in collaboration with Regeneron Pharmaceuticals.
The company said that Zaltrap could be effective in colorectal cancer patients who have not responded to chemotherapy treatment. Large scale clinical trials showed that people taking Zaltrap along with FOLFIRI chemotherapy combination regimen lived for 13.5 months compared to 12 months for those who received only FOLFIRI.
Zaltrap is expected to compete against Roche’s Avastin and Bristol-Myers Squibb’s Erbitux with analysts predicting that it could have peak annual sales of $300 million to $400 million. “While near-term we expect (Regeneron's) stock will continue to trade primarily on the Eylea launch which remains impressive, we view Zaltrap's approval as a nice complement to what is rapidly becoming one of biotech's best commercial stories”, RW Baird analyst Christopher Raymond said.
Source-Medindia