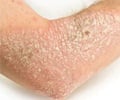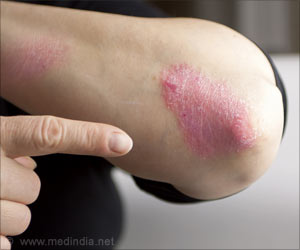
Researchers compared the new drug guselkumab to adalimumab, the most common medication currently used to treat psoriasis. In the trial, 293 adult patients with moderate-to-severe psoriasis (defined as covering 10% or more of the body) were randomly assigned to receive varying doses of one of the two drugs or a placebo over 52 weeks. At weeks 16 and 40, efficacy of both the drugs was measured on a scale of zero to five.
A significantly higher number of patients in the guselkumab group had a score of zero (cleared psoriasis) or one (minimal psoriasis) in both the short and long term periods compared to the adalimumab and placebo groups. At week 40, for example, 81% of patients taking a 200-mg dose of guselkumab had a score of zero or one, compared to 49% of patients taking adalimumab. The study said, "The drug works by blocking a protein specifically implicated in psoriasis, called interluekin 23; older drugs affect the immune process more generally."
An ongoing phase III trial is continuing to test the safety and efficacy of guselkumab as a potential psoriasis treatment.
The study is published in the New England Journal of Medicine.
Source-IANS