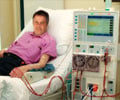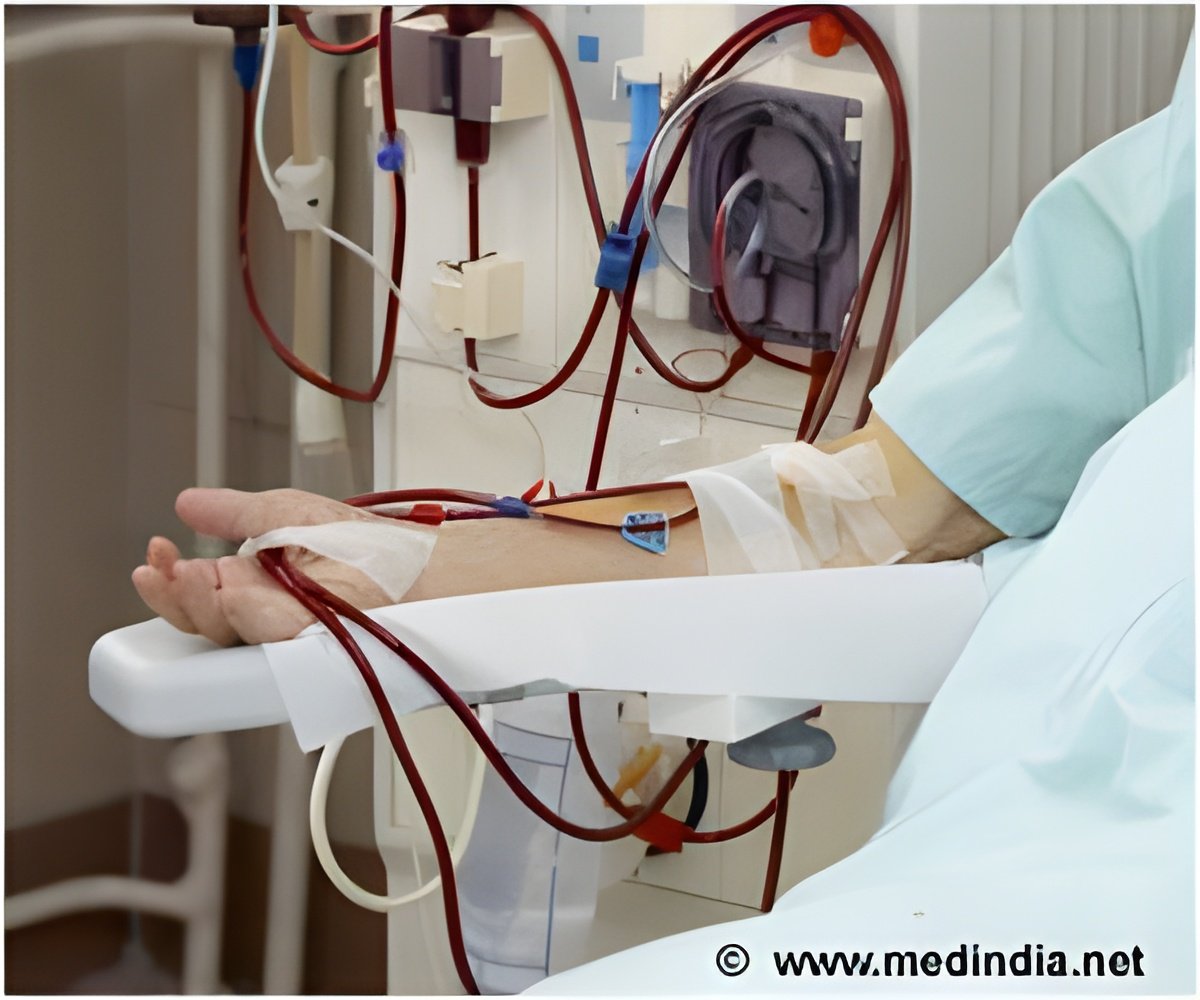
The Tablo System is a consumer friendly device and anyone with guided training can learn how to deliver dialysis.
The number of individual steps the operator has to perform has been reduced by over half compared to existing machines, thanks to various automated processes. The large touch-screen display intuitively leads the user through the dialysis process, with animation and step-by-step instructions.
There’s no need to use distilled water because the system hooks up to the tap and filters it on its own. The system also produces its own dialysis solution, further simplifying the maintenance and operation of the device.
The company is now working toward expanding the FDA indication to include at-home treatment by the patients themselves.
“Dialysis is a life-sustaining therapy that has changed very little in the last few decades. Many patients who receive dialysis want more control over the experience and want to live fuller, more independent lives. These patients could take the lead in their own dialysis care with minimal assistance with our new improved technology. Other areas of medicine suggest that when people are directly engaged in their care, they live longer and feel better,” said Dr. Glenn M. Chertow, M.D, a company official.
Advertisement
Source-Medindia