‘The implantable device called hypoglossal nerve stimulation (HGNS) offers promise for patients with moderate to severe OSA who cannot tolerate CPAP.’
Tweet it Now
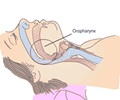
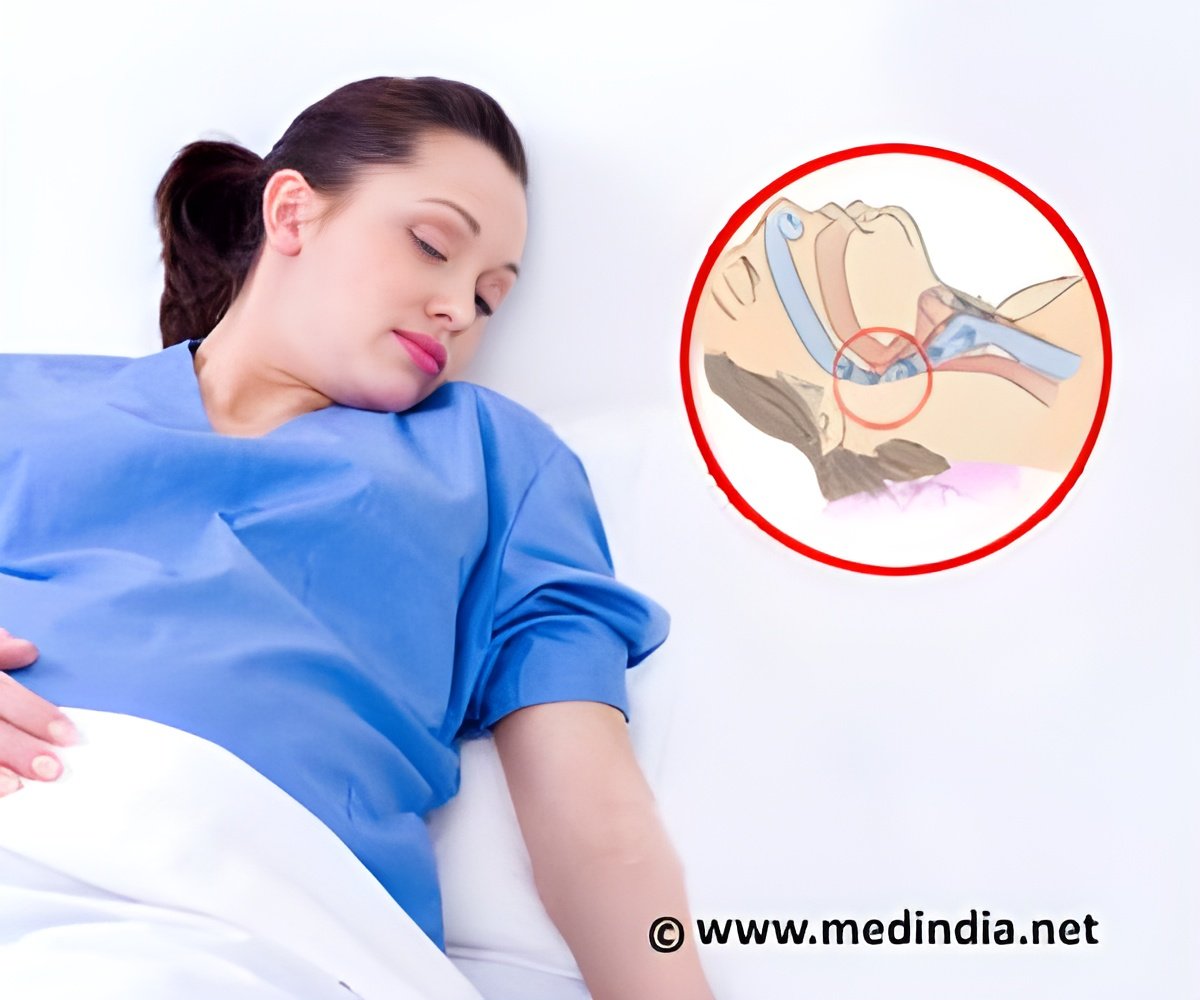
The device is a pacemaker with a tiny generator and a sensing lead, but instead of using electrical pulses to control abnormal heart rate, the device uses two wires to stimulate the tongue. Patients use a remote control to turn on the device before going to sleep and turn it off upon waking up. A delay allows the user to fall asleep before the pulse generator begins stimulation. After detecting the user's breathing pattern, the machine stimulates the hypoglossal nerve (the nerve that controls tongue motion) which enlarges the upper airway. In the current Penn case study, 20 implants were completed at the University of Pennsylvania between January 2015 and March 2016. All patients had information from a baseline polysomnography (PSG) recording prior to HGNS implant, as well as a post PSG approximately two months after HGNS, to assess the severity of their apnea and any change after treatment. Those who received the implant were typically overweight, middle aged, and had severe OSA.
Total apnea-hypopnea index (AHI) - which measures severity of sleep apnea by counting the number of pauses in breathing during sleep - for all patients significantly decreased an average of 35 events per hour after the device was planted, which corresponds to an average reduction of 84 percent. Additionally, the lowest oxygen level measured in the blood during the night significantly increased by 11 percent, from 79 percent to 90 percent.
In the previous STAR clinical trial, NCT01161420, which led to the device's FDA approval in April 2014 for patients unable to tolerate CPAP, patients implanted with the Inspire Upper Airway Stimulation device showed a 78 percent reduction in AHI and 80 percent fewer oxygen desaturation events (number of times the oxygen levels in the blood were low). These results were sustained three years after the device was implanted.
The STAR trial was conducted under controlled conditions, while the Penn study takes the next step by looking at outcomes in post-approval clinical use of the device. By replicating the success of the clinical trial, the findings show that the device is as successful in the general clinical population as it was in participants that met the previous clinical trial criteria.
Advertisement
Source-Eurekalert