First author Paul Kwo, M.D., professor of medicine at the Indiana University School of Medicine, called the results of the international clinical study a landmark achievement."
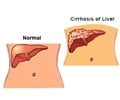
Dr. Kwo, who is medical director of liver transplantation at the IU School of Medicine and IU Health, said that cirrhosis caused by hepatitis C is the leading reason for liver transplantation in the U.S., and those patients have lower survival rates than patients transplanted for other causes of cirrhosis.
Patients with hepatitis C who receive a liver transplant have a 20 to 30 percent chance of developing recurrent cirrhosis within five years after transplant. Hepatitis C, which is usually asymptomatic until severe liver damage occurs, is transmitted by exposure to contaminated blood. The most common way to get hepatitis C is through intravenous drug use, with the Centers for Disease Control and Prevention estimating that 3.2 million individuals in the United States have contracted the hepatitis C virus.
In 2013, 6,400 liver transplants were performed in the U.S., according to the Organ Procurement and Transplantation Network, and nearly 50 percent of those patients had hepatitis C with or without liver cancer.
The phase 2, multi-center trial enrolled 34 liver transplant recipients with hepatitis C who did not have cirrhosis. Of those, 33 patients or 97 percent exhibited no sign of hepatitis C virus at 24 weeks after treatment with the new drug regimen, and none suffered transplant rejection. In a similar phase 3 trial of non-transplant patients with cirrhosis, a historically difficult patient group to treat, the same treatment regimen produced a 96 percent cure rate for hepatitis C.
Advertisement
This international research study was funded by AbbVie. The results from this study and a phase 3, multi-center randomized study will be presented Nov. 11 at the 2014 the American Association for the Study of Liver Disease annual meeting in Boston, Mass.
Advertisement
Source-Medindia