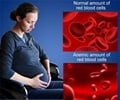
Keeping in mind the escalating incidence of HIV transmission, the WHO has recommended certain important steps for preventing its transmission via mother-to-child and ensuring their survival.
The eradication of HIV transmission from mother-to-child is regarded as a surmounting concern by the WHO and has been included in the millennium development goals as the one of the prime objectives.Both the mitigation of the risk of mother-to-child HIV transmission and the maintenance of the health of the mother and child are of utmost importance and most sought after concerns of the interventions across the globe.
In 2006, various important evidences arose with WHO issuing improvised guidelines on the application of antiretroviral (ARV) prophylaxis to reduce MTCT. It involved period of breast feeding, optimal time to begin antiretroviral therapy (ART) and even the safe techniques of breast feeding for HIV- vulnerable infants.
New improvised recommendations were summarized in 2010 in:Use of antiretroviral drugs for treating pregnant women and preventing HIV infection in infants.
Some of the key recommendations were:-
1. Early recommendation of ART for pregnant females with CD4 cell counts of ≤350 cells/mm3 or who have stage III/IV HIV disease. HIV drugs are also recommended for women who do not require them for their own health but taking these drugs could reduce transmission.
3. Usage of ARVs by breast feeding mothers and infants to prevent the further spread of HIV transmission. Longer duration of treatment in infants is also recommended.
The vital information for the adaptation of WHO recommendations is also mentioned in the detailed guidelines.
The guidelines of 2010 chiefly aim at low- and middle- income segments and target to provide efficient interventions for the reduction of MTCT in a resource-constraint environment.
In breast feeding mothers, MTCT is expected to decline from 35% to 5%( or even lower) and in non-breast feeding mothers from a background threat of 25% to even less than 2%, with the practical introduction of these recommendations.
There is a strong compliance between the WHO 2010 guidelines and the newly recognized WHO guidelines development process. It involves an apt reviewing of the consideration of practicability of the program and the expected implications of the cost pertaining to the application of these recommendations as well as the review of new evidences.
Both the adult ART and HIV and infant feeding guidelines have been updated and revised in a synchronized manner by the WHO.
Sources:-
1. http://www.who.int/hiv/pub/mtct/antiretroviral2010/en/index.html
2. http://www.medscape.com/viewarticle/731135
Source-Medindia