Miravirsen may be an alternative to pegylated interferon and ribavirin in the treatment of chronic hepatitis C.
Chronic hepatitis C infection is one of the major causes of liver failure and liver cancer. The current treatment of hepatitis C with pegylated interferon and ribavirin is associated with serious side effects.A new treatment being tried out for the treatment of chronic hepatitis C infection is called miravirsen. Miravirsen acts by inhibiting "microRNA-122", a protein produced by the liver that interacts with hepatitis C virus; this interaction is important for gene expression and plays a role in the growth and differentiation of the virus, as well as in other vital viral functions. Miravirsen has provided long-lasting viral suppression in animal studies, and has not produced any side effects in initial studies in humans.
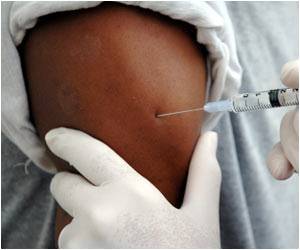
A recent study evaluated the effectiveness of miravirsen in treating patients with chronic hepatitis C liver disease. The patients included in the study were between 18 and 65 years of age and did not undergo any other treatment. The patients received 3 mg, 5mg, or 7mg per kilogram of body weight of miravirsen or a placebo (an inert substance given for the sake of comparison). The injections were administered weekly over one month. Twelve out of the 36 patients in the study received additional treatment with pegylated interferon and ribavirin 7 to 10 weeks after starting miravirsen. The patients were observed for a duration of 18 months.
The study found that the hepatitis C viral load was reduced during administration of miravirsen, and the low levels persisted even after the drug was stopped. During the follow-up period, HCV RNA was not detected in one patient who received 5 mg dosage and in four patients who received 7 mg, indicating a possible cure in these patients. It also reduced cholesterol levels in the patients, indicating an additional beneficial effect
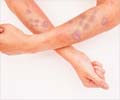
There was no indication that these patients developed resistance to the treatment. However, some patients did show a rebound increase in the viral load after the treatment was stopped. Adverse effects were mild to moderate and did not warrant discontinuation of the treatment. They included headache, infection of the external ear, fainting, injury to the pelvic bone following a fall and injection-site reactions. One patient suffered from loss of consciousness along with a pelvic bone injury after a fall.
Though miravirsen appears to be promising in hepatitis C, larger studies are necessary to establish its benefits and lack of significant adverse effects, before it can become available for treatment of chronic hepatitis C.