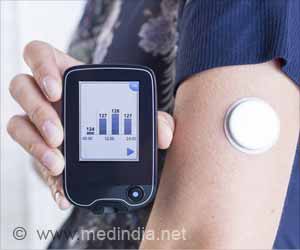
‘The new MiniMed 670G measures sugar levels every 5 minutes and delivers insulin as needed. The device costs between $6,000 and $9,000.’
Tweet it Now
Doctors said they have long awaited a device that could help patients around the clock.
"I can't wait to get my hands on it because I can think of a lot of our patients who need this," said the Cleveland Clinic's Dr. Betul Hatipoglu. "Now I have a helper who is going to help me to help them."Currently, Type 1 diabetes patients manage their insulin through multiple injections throughout the day. They are at increased risk of elevated blood sugar levels, heart disease, and other health complications.
The new MiniMed 670G consists of a drug pump, a sensor that measures blood sugar and a tube that delivers the insulin. The sensor measures sugar levels every 5 minutes, infusing or withholding insulin as needed. Patients have to manually increase insulin before meals.Medtronic said the device would cost between $6,000 and $9,000. The pump is about the size of a deck of cards and can be worn on a belt or carried in a pocket.
Other insulin pumps deliver a baseline level of insulin, and patients monitor their blood sugar levels and inject insulin to keep their blood sugar levels from rising. But the danger is having too much insulin in the body overnight when blood-sugar levels naturally fall. People can go into comas, suffer seizures and even die.
A three-month study was conducted before the FDA approved the device. The trial included more than 120 patients, showing that the device is safe for those 14 years and older.
Advertisement
The device can only be marketed for those with Type 1 diabetes. But eventually, the device can be used by those with Type 2 diabetes.
Advertisement