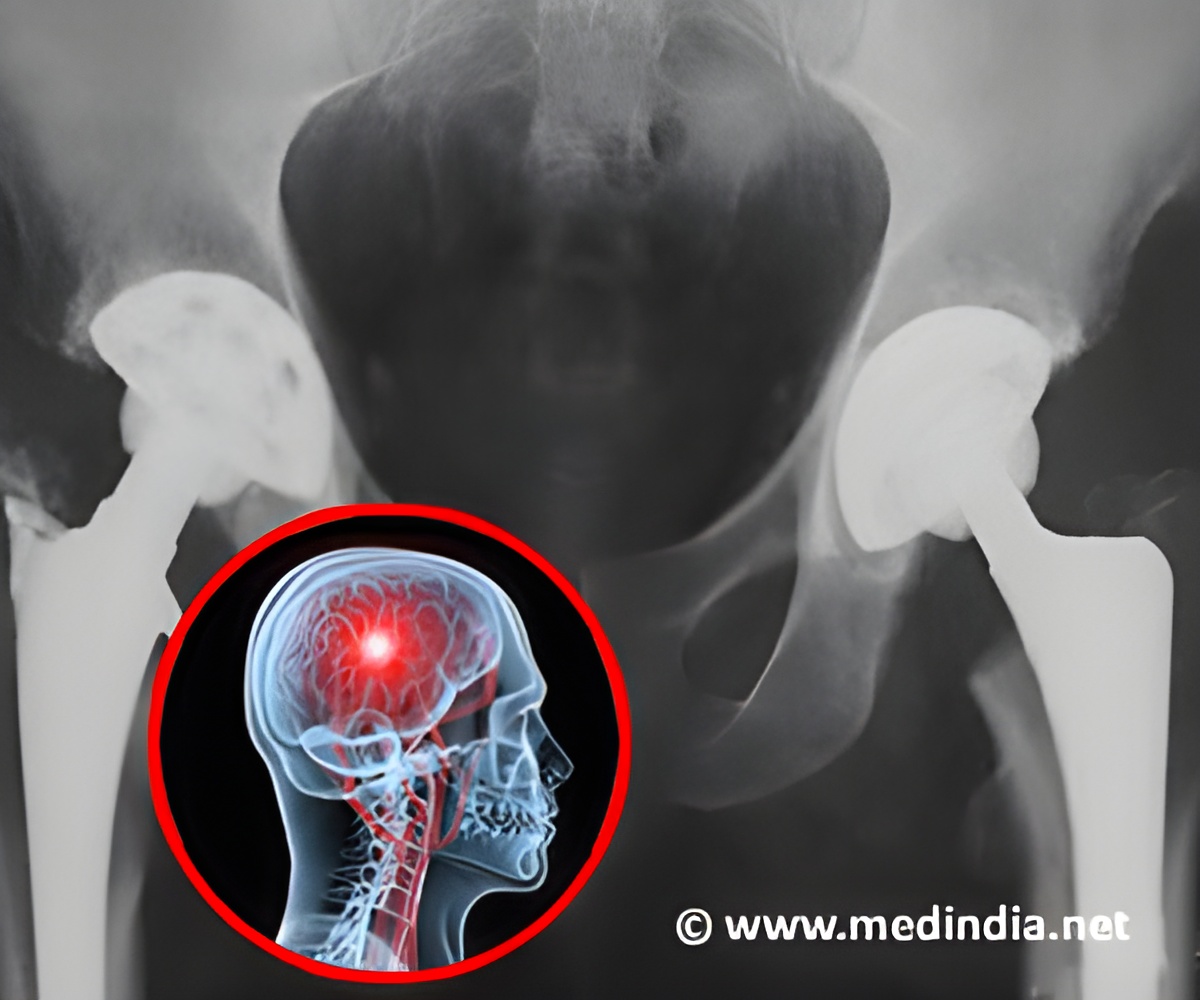
The proposal will close a loophole present in the 1976 federal law under which companies have to only show that their device resembles the ones currently in the market without any need to conduct clinical studies.
The agency has expressed concerns over a large number of all-metal hip implants failing very soon after being implanted, forcing patients to undergo expensive and painful surgeries to replace in implants and also increasing health risks as tiny metallic debris released by the implants often cause bone and tissue damage.
Source-Medindia