‘In trials to date, users have been positive about how use of an artificial pancreas gives them 'time off' or a 'holiday' from their diabetes management, since the system is managing their blood sugar effectively without the need for constant monitoring by the user.’
Tweet it Now
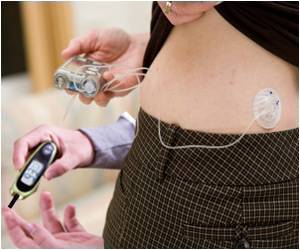
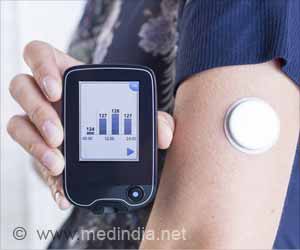
Currently available technology allows insulin pumps to deliver insulin to people with diabetes after taking a reading or readings from glucose meters, but these two components are separate. It is the joining together of both parts into a 'closed loop' that makes an artificial pancreas, explain authors Roman Hovorka and Hood Thabit of the University of Cambridge, UK. One part of the clinical need for the artificial pancreas is the variability of insulin requirements between and within individuals, on one day a person could use one third of their normal requirements, and on another 3 times what they normally would. This is dependent on the individual, their diet, their physical activity and other factors.
The combination of all these factors together places a burden on people with type 1 diabetes to constantly monitor their glucose levels, to ensure they don't end up with too much blood sugar (hyperglycaemic) or more commonly, too little (hypoglycaemic). Both of these complications can cause significant damage to blood vessels and nerve endings, making complications such as cardiovascular problems more likely.
The actual timeline to availability of the artificial pancreas, as with other medical devices, encompasses regulatory approvals with reassuring attitudes of regulatory agencies such as the US Food and Drug Administration (FDA), which is currently reviewing one proposed artificial pancreas with approval possibly as soon as 2017. And a recent review by the UK National Institute of Health Research (NIHR) reported that automated closed-loop systems may be expected to appear in the (European) market by the end of 2018.
They concluded: "Significant milestones moving the artificial pancreas from laboratory to free-living unsupervised home settings have been achieved in the past decade. Through inter-disciplinary collaboration, teams worldwide have accelerated progress and real-world closed-loop applications have been demonstrated. Given the challenges of beta-cell transplantation, closed-loop technologies are, with continuing innovation potential, destined to provide a viable alternative for existing insulin pump therapy and multiple daily insulin injections."
Advertisement