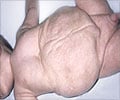
The acquisition gives Amicus access to Scioderm’s experimental topical cream, Zorblisa, for the treatment of epidermolysis bullosa, a rare connective tissue disease with no approved treatment yet.
“EB is a disorder that is utterly devastating and painful as it causes extremely fragile skin that blisters and tears from minor friction or trauma. In many children it leads to severe complications and a very early death. We believe we are well-positioned to rapidly complete the clinical development of Zorblisa and to make Zorblisa commercially available for all EB patients as quickly as possible,” said Crowley.
Crowley has described Zorblisa’s market as worth “a billion dollars-plus." The existing palliative treatments consisting of bandaging and pain management for EB patients cost $10,000-15,000 per month.
Scioderm CEO Robert Ryan said the acquisition of his company will provide additional expertise and resources to allow Zorblisa to come to market.
Zorblisa is currently in Phase III trials. In 2013, the drug received a Breakthrough Therapy designation from the US Food and Drug Administration, after early trials showed the drugs ability to help wounds across all EB subtypes heal.
Advertisement
Source-Medindia