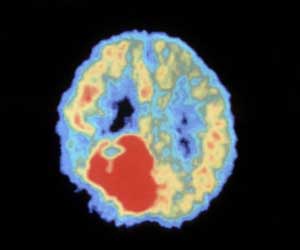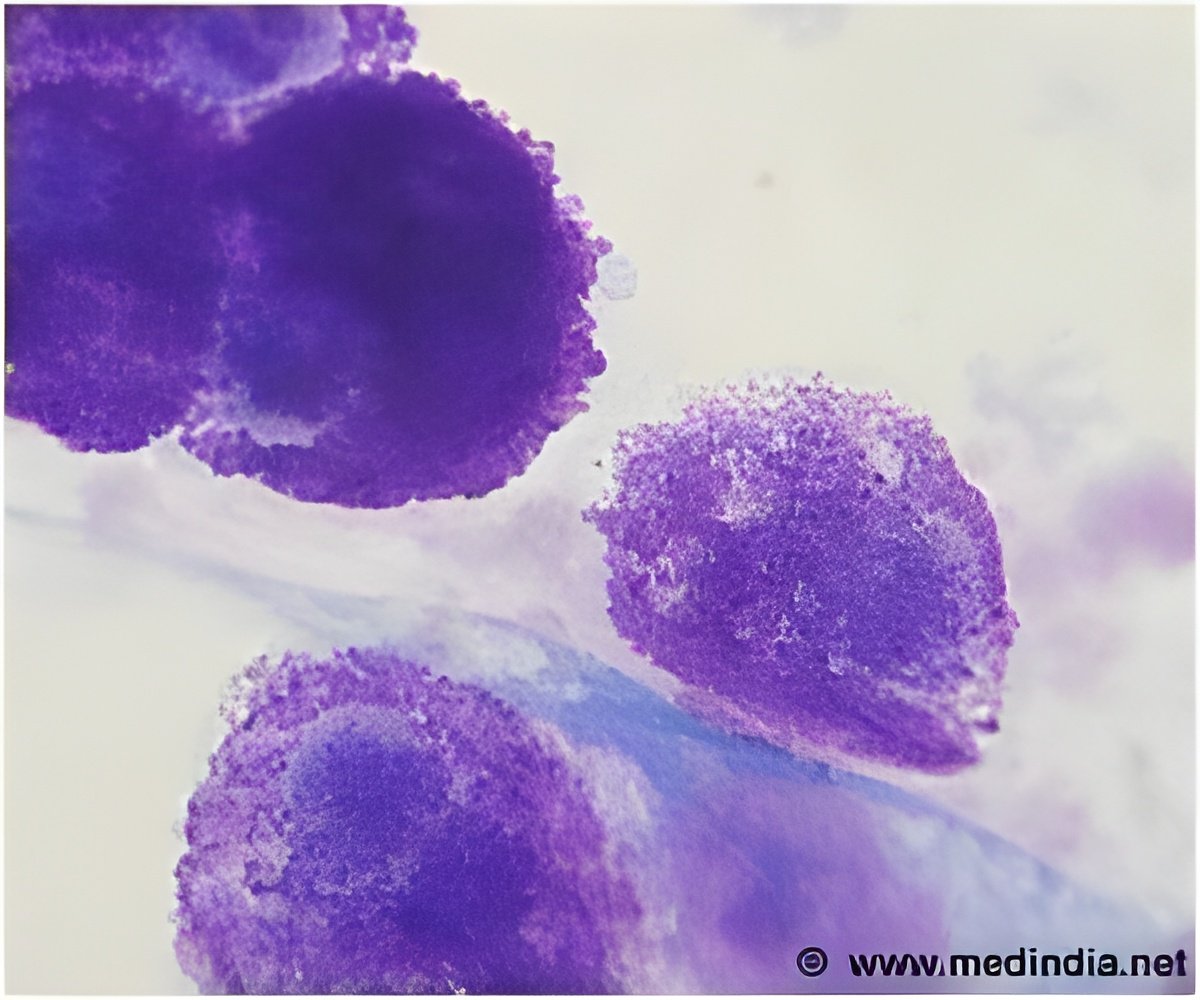
‘A new nano-fabricated platform allows brain cancer cells to be observed individually, and isolates a subset of particularly aggressive cells. This small group of cells plays a critical role in what makes glioblastoma such a deadly form of cancer.’
Tweet it Now
By creating an environment similar to the one that glioblastoma cells naturally navigate, Yale and Hopkins researchers could accurately predict the clinical outcomes of the 14 brain cancer patients enrolled in a recent study. The results are published in the journal Cell Reports. Prof. Andre Levchenko, director of Yale's Systems Biology Institute and John C. Malone Professor of Biomedical Engineering, said the platform allows cells to be observed individually, and isolates a subset of particularly aggressive cells. This small group of cells plays a critical role in what makes glioblastoma such a deadly form of cancer.
"We can now analyze all these effects on the single-cell level, which is important because there are a multiple cell subpopulations in the tumor," said Levchenko, a co-senior author of the study. "A tumor is a community of cells - not just one type of cell."
Conversely, genetic analyses look at many different cells at once, and crucial details are lost. As a result, these tests offer scant information for predicting patient outcomes.
To create the appropriate environment, the researchers engineered a polymeric nanostructure that mimics certain aspects of brain tissue architecture. Unlike a flat and hard Petri dish, this environment provides cells with a fibrous structure.
Advertisement
"Surprisingly, we could predict how many months would pass before a specific tumor would recur by observing how the cells moved after being surgically removed," Levchenko said.
Advertisement
For a comparison, the researchers also observed cells placed in a Petri dish. In that environment, the glioblastoma cells were much more passive and unresponsive to changes in chemical cues, including PDGF. This underscores the importance of placing patient cells into an appropriate environment for diagnostic and prognostic testing.
Besides developing a more precise prognosis, the researchers believe the technology could lead to new therapies and better match existing ones to specific patients, a goal of Yale Systems Biology Institute.
The initial analysis of the 14 patients was conducted at Johns Hopkins, and the platform is now being integrated into clinical settings at Yale, Johns Hopkins and the Mayo Clinic.
Source-Newswise