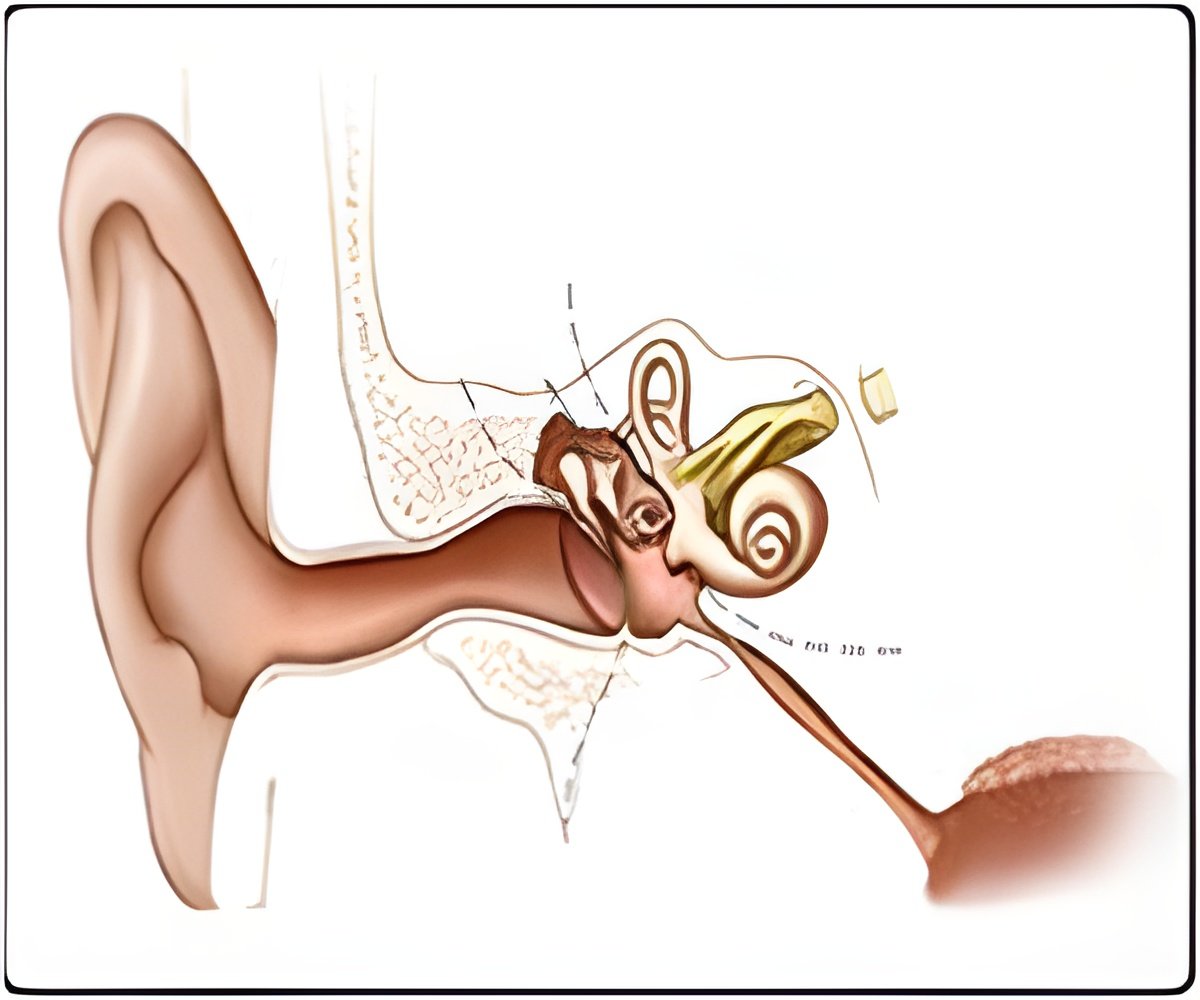
The mutation (called R156W), was first identified in an individual suffering from cochlear hearing loss, and it affects the way the myo1c protein interacts with proteins known as actin filaments, another crucial component of the sensory apparatus of the inner ear.
Michael Greenberg and his colleagues at UPenn have examined the biochemical and mechanical properties of the mutant myosin protein. Comparing constructs of the normal, "wild-type" protein to the R156W mutant, they examined the two proteins' kinetics and motility and discovered the mutant has a reduced sensitivity to mechanical loads and a lower duty ratio, meaning it spends less time attached to actin filaments.
Though the cochlear cell myo1c defects are associated with hearing loss, how this mutation causes the disease is still a mystery. The exact molecular role of myo1c is hazy, although it has been linked to several important cellular processes including hearing and insulin stimulated glucose uptake within cells.
Understanding the defects caused by the R156 mutation could help to solve the puzzle.
"R156 is a highly conserved residue throughout the myosin superfamily. The fact that mutation of this residue affects the myosin duty ratio and strain sensitivity may very well be applicable in other myosins as well. In the long term, we hope to gain greater insight into the mechanism of myosin strain sensitivity and its role in mechanotransduction," said Greenberg.
Advertisement