DES was widely used in 1940s to prevent miscarriage, premature birth and pregnancy complications.
Research in the 1950s showed that DES didn't prevent these complications, and in 1971, the drug was banned after research found that exposure to DES in the womb raised the risk of a rare form of vaginal cancer among daughters of women who took the drug.
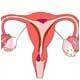
For the new study, the researchers studied 6,500 women (4,600 exposed to DES) and found the daughters with exposure to DES in the womb had an increased risk of 12 medical conditions.
They were also twice as likely to be infertile and had a five-fold increased risk of having a pre-term delivery.
The women also had 40 times the risk of developing a rare cancer of the vagina among young women, called clear cell adenocarcinoma - although the number of cases remained about one in 1000.
Advertisement
"Our study carefully documents elevated risk for DES-exposed daughters for a host of medical problems-many of them also quite common in the general population," said study author Robert N.
Advertisement
"Without the sentinel finding of a very rare cancer in young women, and without the sustained follow-up of those who were exposed, we would not know the full extent of harm caused by DES exposure in the womb," he added.
The study, however, did not evaluate sons with DES exposure in the womb, but previous reports have indicated an increased risk for certain testicular abnormalities.
The results were published Oct. 6, 2011, in the New England Journal of Medicine.
Source-ANI