Drug manufacturer Dr Reddy's Laboratories has revealed that it has received approval from USFDA for its Abbreviated New Drug Application for Omeprazole Mg OTC.
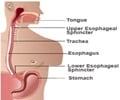
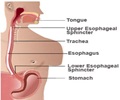
This was revealed in a filing with the Bombay Stock Exchange. The company said that the formulation contains 20.6 mg Omeprazole Mg and the dosage form is a capsule.The drug is indicated in the management of heartburn and is to be taken orally.
Source-Medindia
RAS