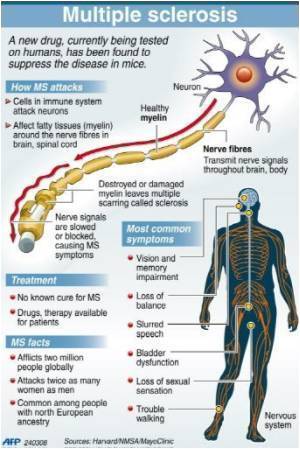
The study involved 1,106 people with relapsing-remitting MS in 24 countries. The participants received either a once-daily oral dose of 0.6 milligrams of laquinimod or a matching placebo for two years. Eighty percent of those taking laquinimod and 77 percent of those taking the placebo finished the two-year study.
Patients treated with laquinimod experienced a statistically significant reduction of 23 percent in annual relapse rate, compared to patients treated with a placebo. Additionally, there was a reduction of 36 percent in disability progression, as well as a 33 percent reduction in brain atrophy for those people treated with laquinimod.
“These exciting results confirm that laquinimod has a significant impact on progression of disability and disease activity, while maintaining a high safety profile,” said lead author Giancarlo Comi, MD, director of the Department of Neurology and Institute of Experimental Neurology at the Scientific Institute and University Vita-Salute San Raffaele in Milan, Italy. “This may be attributed to the novel mechanism of action of laquinimod, which effectively and safely addressed both the acute inflammatory activity and the accumulation of irreversible tissue damage. This suggests a substantial future role for laquinimod in the treatment of MS.”
Laquinimod was safe and well tolerated. Overall frequencies of adverse events were low and comparable to those observed in the placebo group. “The incidence of liver enzyme elevation was higher in laquinimod treated patients,” said Comi. “However, these elevations were temporary, reversible and did not lead to any signs of liver problems.”
The study was supported by Teva Pharmaceuticals.
Advertisement
Source-Medindia