Dengue affects between 50 and 100 million people in the tropics and subtropics each year, causing fever, muscle and joint ache as well as potentially fatal dengue haemorrhagic fever and dengue shock syndrome.
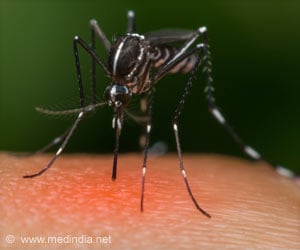
The disease is caused by four strains of virus that are spread by the mosquito Aedes aegypti. There is no vaccine, which is why scientists are focussing so intensely on mosquito control.
In 2009, Turelli and others hit on the idea of inserting a naturally-occurring bacterial parasite called Wolbachia to shorten the mosquito's lifespan so that the virus would not have enough time to develop.
Initial excitement was followed by disappointment. The strain of Wolbachia they used was somewhat virulent and knocked out the mosquitoes before they had the chance of spreading into the wild mosquito population.
Going back to the drawing board, the scientists found a non-virulent strain of Wolbachia in the fruitfly Drosphila -- a standard choice for laboratory research -- and believed they had the answer.
Advertisement
Yet it was also harmless. The mosquito's fitness, a measure of its ability to survive and reproduce, was reduced only by about 10 to 20 percent.
Advertisement
It lives inside cells and is maternally inherited, thus raising the possibility that after a few generations, the introduced dengue-free mosquitoes eventually outnumber dengue-carrying counterparts.
After long consultations with the government and regulators, the investigators released hundreds of thousands of Wolbachia-infected mosquitoes in two locations in Queensland, Australia, this year.
Early results suggest that the introduced mosquitoes have thrived, reaching 100 percent of mosquitoes that were captured and analysed in one location and more than 80 percent in the other. Some were also spotted several kilometers (miles) beyond the release area.
The experiments "herald the beginning of a new era in the control of mosquito-borne diseases," said Jason Rasgon, a specialist at the Johns Hopkins Malaria Research Institute, Maryland, in a commentary also carried by Nature.
"The advantage of population-replacement approaches is that, once established, they are self-propagating. And because the mosquito population is simply changed rather than eliminated, effects on the ecosystem should be minimal."
The paper appears in Nature, the weekly British science journal.
Scientists are now hoping the trial will now be rolled out overseas.
"The next step for us in Australia will be continuing to monitor the field experiments that we have and also some expansion of those in the next wet season," said Professor Scott O'Neill from Monash University.
"Most importantly, we're wanting to move to areas in the world where we have much stronger levels of dengue transmission like Thailand, Vietnam, Brazil and Indonesia."
Source-Medindia