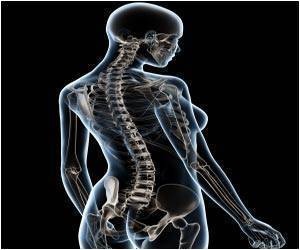
Removal of the cervical disc relieves pain by eliminating friction between the vertebrae and/or nerve compression. Spinal fusion is used following surgery for degenerative disc disease, where the cusioning cartilage has worn away, leaving bone to rub agains bone and herniated discs, where the discs pinch or compress nerves.
"We hope that this investigational procedure eventually will help those who undergo spinal fusion in the back as well as in the neck," said Kim, who also is chief of spinal neurosurgery at UC Davis. "And the knowledge gained about stem cells also will be applied in the near future to treat without surgery those suffering from back pain."
Millions of Americans are affected by spine diseases, with approximately 40 percent of all spinal fusion surgery performed for cervical spinal fusion. Some 230,000 patients are candidates for spinal fusion, with the numbers of potential patients increasing by 2 to 3 percent each year as the nation's population ages.
"This is an exciting clinical trial to test the ability of the bone-forming stem cells from healthy donors to help patients with spinal disease," said Jan Nolta, director of the UC Davis Institute for Regenerative Cures.
"For the past 50 years, bone marrow-derived stem cells have been used to rebuild patients' blood-forming systems. We know that subsets of stem cells from the marrow also can robustly build bone. Their use now to promote vertebral fusion is a new and extremely promising area of clinical study," she said.
Advertisement
In the surgery, called an anterior cervical discectomy, a cervical disc or multiple discs are removed via an incision in the front of the neck. The investigational stem cell therapy then is applied to promote fusion of the vertebrae across the space created by the disc removal.
Advertisement
Adequate spinal fusion fails to occur in 8 to 35 percent or more of patients, and persistent pain occurs in up to 60 percent of patients with fusion failure, which often necessitates additional surgery.
"A lack of effective new bone growth after spine fusion surgery can be a significant problem, especially in surgeries involving multiple spinal segments," said Schrot, co-principal investigator for the study. "This new technology may help patients grow new bone, and it avoids harvesting a bone graft from the patient's own hip or using bone from a deceased donor."
Current methods of promoting spinal fusion include implanting bone tissue from the patient's hip or a cadaver to encourage bone regrowth as well as implanting bone growth-inducing proteins. However, the Food and Drug Administration has not approved the use of bone morphogenetic proteins for cervical spinal fusion. Their use has been associated with life-threatening complications, particularly in the neck.
The leading-edge stem cell procedure is part of a prospective, randomized, single-blinded controlled study to evaluate the safety and preliminary efficacy of an investigational therapy: modified bone marrow-derived stem cells combined with the use of a delivery device as an alternative to promote and maintain spinal fusion.
The study includes 10 investigational centers nationwide. The UC Davis Department of Neurological Surgery anticipates enrolling up to 10 study participants who will be treated with the stem cell therapy and followed for 36 months after their surgeries. A total of 24 participants will be enrolled nationwide.
Source-Eurekalert