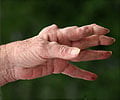
Rheumatoid arthritis sufferers as well as activists are expressing their anger over a decision of health watchdog NICE. The organization announced that a drug touted to ease an "aggressive" form of arthritis would not be approved for use on the NHS.
The National Institute for Clinical Excellence (NICE) gives that it will not reconsider its recommendation not to prescribe abatacept (or Orencia) for the treatment of moderate to severe rheumatoid arthritis. In spite of Arthritis charities campaigning against the motion, NICE issued final draft guidance recommending against its use. The guidance will be applied to the NHS later this year, if no further appeals are heard."The independent advisory committee reviewed the evidence on abatacept, taking into account information on the nature of the condition and consultation responses from people with rheumatoid arthritis, patient/carer groups and clinical specialists”, a NICE spokesperson was quoted.
Recently, NICE recommended rituximab for rheumatoid arthritis. Evidence shows that abatacept can cost twice as much as rituximab, but offer only similar benefits.
"Whilst recognizing the severity of the disease, the Committee concluded that based on this evidence, abatacept could not be considered a good use of NHS resources. However, the draft recommendations also say that people currently receiving abatacept for the treatment of rheumatoid arthritis should have the option to continue therapy until they and their clinicians consider it appropriate to stop”, the report from NICE read.
"We strongly believe that this is the wrong decision for people living with severe rheumatoid arthritis in the UK and a potential step backwards in the management of the condition.
"These patients will have already tried other treatments for rheumatoid arthritis and failed on them over time. To deny patients abatacept is to deny people with a very aggressive and severe form of rheumatoid arthritis one more chance at controlling the disease and condemn them to a life of pain and disability.
Advertisement
"Palliative care and the burden of ongoing disability will result in significantly greater costs for the NHS than a treatment which can make a significant difference to the quality of life of people with severe rheumatoid arthritis”, Ailsa Bosworth, chief executive of The National Rheumatoid Arthritis Society (NRAS) was quoted.
Advertisement
Meanwhile Abbott Laboratories’ Humira , Amgen Inc's Enbrel and Johnson & Johnson unit Centocor's Remicade were the rheumatoid arthritis drugs recommended on Wednesday for use in Britain's state health service.
Source-Medindia
ANN/V