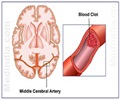
Neurobiological Technologies, Inc. (NTI) announced today the presentation of new analysis of data from prior clinical trials of ancrod and updates on the clinical development of their investigational drug, Viprinex(TM) (ancrod), for the treatment of acute ischaemic stroke at the 16th annual European Stroke Congress in Glasgow, Scotland in the United Kingdom. Viprinex is a defibrinogenating agent derived from the venom of the Malayan pit viper.
"The ancrod data presented this week in Glasgow draws positive conclusions that support a new dosing regimen for ancrod and revised inclusion/exclusion criteria for the current clinical trials," said David Sherman, MD, University of Texas Health Science Center at San Antonio, USA."The reanalysis enables us to see that ancrod may be safe and effective when administered to patients within six hours of stroke onset. With so few treatments currently available to stroke patients, we look forward to the results of the international phase 3 trials. We hope the phase 3 results provide evidence of definitive safety and efficacy of ancrod in ischaemic stroke in the six-hour time window."
NTI is moving forward with two international, randomized, double-blind, placebo-controlled phase 3 trials for Viprinex(TM) as part of the Ancrod Stroke Program (ASP). These trials, known as ASP-I and ASP-II, will evaluate whether a brief (2 or 3 hour) infusion of ancrod begun up to six hours after onset of stroke can reduce stroke related disability. ASP-I and II are currently enrolling and each trial is planned to enroll 650 patients. Patient accrual began in November 2005.
The trials are being conducted in the U.S., Europe, South Africa, Australia, and New Zealand. Additional countries will be entering the program. The company expects that the trials will be conducted at up to 130 sites worldwide and that approximately 70% of the ASP-I sites and approximately 50% of the ASP-II sites will be located within the U.S. Patients who qualify for enrollment into the clinical trials will receive a two or three-hour intravenous infusion of ancrod or placebo within six hours of stroke onset. If treated within three hours of stroke onset, subjects must have been determined unsuitable for treatment with tPA.
"We have data showing that fibrinogen plays a significant role in influencing stroke outcome. Ancrod directly targets fibrinogen and has the potential to treat strokes by rapidly lowering fibrinogen levels," said David E. Levy, MD, Vice President of Clinical Development for NTI.
"We are aggressively recruiting patients worldwide for the ASP-I and ASP-II studies. We hope by garnering a large patient base for the study we can generate the robust data needed to confirm our new dosing strategy and six hour treatment window for acute ischaemic stroke."
Advertisement
LIN/M