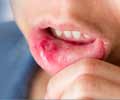
"The vaccine has been shown to be very effective to reduce the risk for shingles infection in older adults and is currently recommended for all older patients who are not receiving immunosuppressive medications, explains the study''s lead investigator, Jeffrey Curtis, MD, MPH; assistant professor, Division of Clinical Immunology and Rheumatology, University of Alabama at Birmingham. "The concern for people who are taking immunosuppressive medications, such as patients with rheumatoid arthritis taking anti-TNF therapies [e.g. Enbrel®, Humira®, Remicade®, Cimzia®, Simponi®] is that since the vaccine contains a weakened version of the live virus, it could allow the virus to reactivate and cause a short-term increase in the risk for shingles infection - within six weeks of vaccination. Beyond that initial time period, the vaccine would likely decrease the risk for shingles infection by helping the body form natural protective antibodies to prevent the infection."
Using data from the U.S. Medicare program in 2006 to 2009, Dr. Curtis'' team identified 6,793 patients with autoimmune diseases who were over the age of 60 and who received the live zoster vaccine. The majority were Caucasian women with an average age of 74-and-a-half. Seventy-six vaccinated patients had ankylosing spondylitis; 1,260 had inflammatory bowel disease; 161 had psoriatic arthritis; 1,745 had psoriasis; 3,246 had rheumatoid arthritis; and 305 had two or more of these diseases.
At the time of vaccination, or within the previous 60 days, 580 patients were taking biologics (mostly anti-TNF therapy); 1,825 were taking disease-modifying anti-rheumatic drugs (commonly called DMARDS); and 1,432 were taking steroids (such as prednisone).
There were 387 people vaccinated who were current users of biologic medications and not steroids. Among these people, there were no cases of shingles in the six weeks following vaccination. Another 193 people on biologics and steroids were vaccinated, and their rate of the shingles infection was no higher than that of vaccinated people taking non-biologic DMARDs and steroids, but no biologics.
"In both of these groups taking steroids, only about five out of 1,000 people [less than one percent] developed the infection," says Dr. Curtis. "Steroids have been previously shown to significantly increase the risk for shingles infection, so this finding was not a surprise."
These findings led the researchers to conclude that people taking biologics may be able to receive the zoster vaccine safely and may not be at an increased, short-term risk of developing shingles from the vaccine. Dr. Curtis and his team have received funding through the ACR Research and Education Foundation''s Within Our Reach campaign to continue this research as a clinical trial in hopes of demonstrating the safety of the live zoster vaccine in people receiving biologic medications.
Advertisement
Advertisement
Editor''s Notes: Jeffrey R. Curtis, MD, MPH and his colleagues, including Dr. Sophie (Jie) Zhang, will present this research during the ACR Annual Scientific Meeting at McCormick Place Convention Center at 5:45 PM on Sunday, November 6 in Room W470b. Dr. Curtis will be available for media questions and briefing at 8:30 AM on Tuesday, November 8 in the on-site press conference room, W 175 C.
Learn more about living well with rheumatic disease as well as rheumatologists and the role they play in health care. Also, discover the ACR''s Simple Tasks campaign, which highlights the severity of rheumatic diseases and the importance of early and appropriate referral to a rheumatologist.
Presentation Number: 806
Safety of Zoster Vaccine in Individuals with Rheumatoid Arthritis, Psoriatic Arthritis, Ankylosing Spondylitis, and Inflammatory Bowel Disease
Jeffrey R. Curtis (University of Alabama at Birmingham, Birmingham, AL)
Jie Zhang (University of Alabama at Birmingham, Birmingham, AL)
Fenglong Xie (University of Alabama at Birmingham, Birmingham, AL)
Elizabeth S. Delzell (University of Alabama at Birmingham, Birmingham, AL)
Lang Chen (University of Alabama at Birmingham, Birmingham, AL)
Kenneth G. Saag (University of Alabama at Birmingham, Birmingham, AL)
Kevin L. Winthrop (Oregon Health Science Univ, Portland, OR)
Background/Purpose: The live zoster vaccine is approved in the US for individuals ages 50 and older to reduce risk and severity of Herpes zoster infection, also known as shingles. Based upon limited data, the vaccine is currently considered to be contraindicated in individuals taking biologics. The objective of the study is to examine whether the administration of zoster vaccine is associated with an increased risk of Herpes zoster shortly following vaccination among persons treated with biologics for autoimmune and inflammatory diseases.
Method: From 2006 through 2009, we identified from the 100% Medicare fee for service population individuals who were 60 years of age or older; had rheumatoid arthritis, psoriasis, psoriatic arthritis, ankylosing spondylitis, or inflammatory bowel disease; and received zoster vaccine. We classified these individuals according to their concomitant medications (biologics; non-biologic DMARDs, including methotrexate, leflunomide, hydroxychloroquine, and sulfasalazine; oral glucocorticoids) within 60 days prior to or after vaccination. The cumulative incidence of Herpes zoster infection within 42 days following vaccination (the typically-used risk window in zoster vaccine trials) was calculated according to drug exposure.
Result: A total of 6793 subjects were vaccinated; among these, 76 had ankylosing spondylitis, 1,260 had inflammatory bowel disease, 161 had psoriatic arthritis, 1,745 had psoriasis, 3,246 had rheumatoid arthritis, and 305 had two or more diseases; 580 were on biologics, 1825 on non-biologic DMARRDs, and 1432 were on glucocorticoids at the time of or immediately prior (+- 60 days) to vaccination. The mean ± standard deviation (SD) age of vaccinated patients was 74.5 ± 6.3, 72% were women, and 96% were Caucasians.
Within 42 days following vaccination, the overall cumulative incidence of Herpes zoster infection was 2.2 per 1,000 persons. The cumulative incidence of zoster infection was not significantly different between those who were using biologics (1.7 per 1,000) or only non-biologic DMARDs (2.7 per 1,000). Irrespective of biologic or DMARD use, the short term risk of zoster infection was higher for patients receiving oral glucocorticoids compared to those not (p = 0.07) (Table).
Conclusion: Zoster vaccine in current biologic users does not appear to be associated with a short term risk of Herpes zoster infection following vaccination compared to those using only non-biologic DMARDs. In addition, the risks were not different than the rates observed in the general population participating in a large zoster vaccine trial of 38,546 individuals in which the cumulative incidence of Herpes zoster infection within 42 days 1.2 per 1,000 among those who receiving placebo. Vaccination with the live zoster vaccine may be reasonable to consider for patients currently using biologics.
Disclosure: J. Zhang, Amgen, 2 ; F. Xie, None; E. S. Delzell, Amgen, 2 ; L. Chen, None; K. G. Saag, None; K. L. Winthrop, None; J. R. Curtis, Pfizer, BMS, Crescendo, Abbott, Roche, Genetech, UCB, Centacor, Corrona, Amgen, 5
View the full abstract at www.rheumatology.org
Source-Newswise