In a medical first, dialysis patients have been successfully fitted with blood vessels engineered solely from their own living tissue, according to a study published Friday.
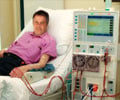
Tested on ten patients, the grafts could become a stable and inexpensive solution to a common problem in dialysis treatment, and were hailed as a "milestone" by researchers not involved in the trials.To compensate for kidney failure, most dialysis patients have their blood filtered three times a week to remove wastes and excess fluid from the body.
For the procedure to work, a short bypass, or shunt, must be inserted between an artery and a vein. In some cases, this additional bit of arterial plumbing can be taken from an existing vein.
In most patients, however, this is not possible, and the bridge has to be made from plastic, which has a significantly higher rate of failure.
The study, published in the British medical journal The Lancet, focused on ten adult patients from kidney treatment centres in Buenos Aires, Argentina and Katowice, Poland whose earlier grafts had given out or who were candidates for plastic tube graft.
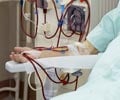
All were fitted with new shunts grown in a laboratory from their own adult cells.
Advertisement
The resulting tube-like membrane is then encased with another sheet of living cell tissue, and implanted in the patient.
Advertisement
A fourth patient died and a fifth was withdrawn, both for reasons unrelated to the trials.
The five remaining patients had grafts functioning for dialysis for between six and 20 months after implantation, with only one needing surgical correction.
"This study demonstrated that we can provide the requisite mechanical strength in a tissue-engineered construct without relying on synthetic biomaterials," concluded the study, led by Todd McAllister of Cytrograft Tissue Engineering in Novato, California.
"The fact that our long-term intervention rate was so low -- even with this high risk group -- offers hope to patients suffering from end-stage renal disease."
There are between 1.5 and 2.0 million people receiving dialysis treatment around the world, and many millions more in need, according to epidemiological studies.
In a commentary, also published in The Lancet, Vladimir Mironov of the Medical University of South Carolina and Vladimir Kasyanov of Riga Stradins University in Latvia said the cost of the innovative technique would need to come down before it was commercially viable.
But they hailed the clinical tests as a "revolutionary milestone," noting that developments in nanotechnology opened "realistic opportunities for reduction of industrial production time and cost."
Source-AFP
LIN