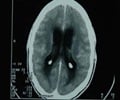
There is no urgency for vaccinating children against meningitis as the prevalence is relatively low, say Canadian health authorities.
At the same experts have taken strong objection to a TV commercial promoting the vaccine Menactra manufactured by Sanofi Pasteur. The commercial shows what might happen if parents don't vaccinate their children but critics warn parents to examine the facts and not be swayed by such ads.'The [TV] ads for the vaccine I've seen are very problematic,' Dr. Barbara Mintzes, a member of the Drug Assessment Working Group at the University of British Columbia, told CBC News. 'I find it really a problem because of the way it's playing into that parental concern to protect their child and using that to sell a product.'
Menactra protects against four strains of meningitis-causing bacteria.
On Oct. 18, the U.S. Food and Drug Administration allowed Sanofi Pasteur, to expand its use to children two to 10 years of age, in addition to the current age indication of 11 to 55 years.
But the Public Health Agency of Canada's National Advisory Committee (PHAC) on Immunization says that given the low incidence of the strains of meningitis covered by Menactra, it feels mass inoculations aren't necessary.
It decided in May not to recommend the vaccine for children aged two to 10 for coverage under provincial health plans, although Prince Edward Island and New Brunswick regions have decided to add Menactra to their childhood immunization schedules.
Advertisement
'Combined, we see an average of about 37 cases a year in Canada' of the three added strains Menactra covers, Dr. Bryna Warshawsky, the immunization committee's co-chair, told CBC News.
Advertisement
Many may not know, say experts, is that the vaccine has side-effects, such as links to Guillain-Barré syndrome. According to the CDC, between March 2005 and September 2006, 17 cases of the syndrome occurred in the U.S. in 11- to 19-year-olds who had received Menactra.
Guillain-Barré syndrome is a serious neurological disorder in which the body's immune system attacks part of the peripheral nervous system. Symptoms include weakness or tingling sensations in the legs, which can spread to the arms and upper body. These symptoms can increase until a person is almost totally paralyzed, although the majority of patients do recover.
Sanofi Pasteur does warn about the risks on its website.
'Guillain-Barré syndrome [GBS] has been reported in temporal relationship following administration of Menactra vaccine. Persons previously diagnosed with GBS should not receive Menactra vaccine.'
The company said the vaccine is safe and that it doesn't understand the immunization committee's position.
'Sanofi Pasteur was disappointed to learn that [the committee] failed to recommend the use of Menactra for the younger group,' the company said in a statement. 'Fatal cases of [meningitis strains], which are vaccine-preventable, continue to be reported in Canada. We believe the broadest protection possible against [meningitis] is essential in Canada.'
Bryna Warshawsky of the PHACsaid there's been no increase in the incidence of rare forms of meningitis in Canada, which would warrant a review of the public health agency's decision.
'Right now, we don't see an increased prevalence,' she said, adding that the immunization committee's position could change.
Source-Medindia
GPL/V