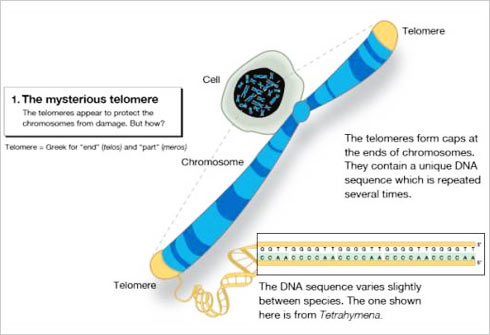
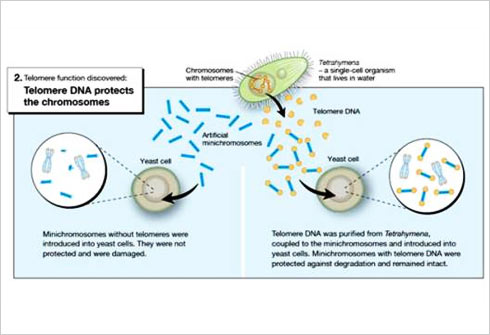
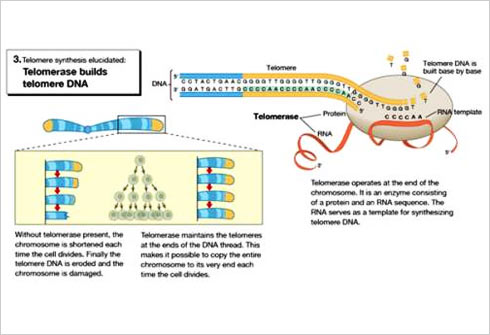
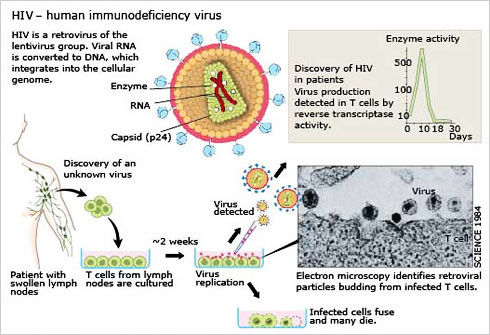
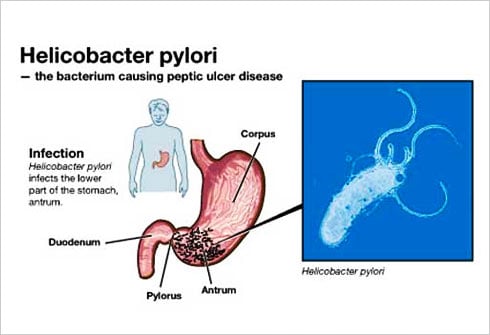
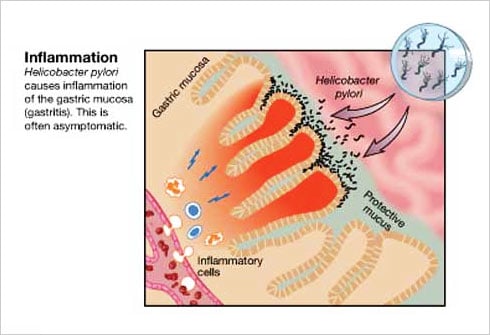
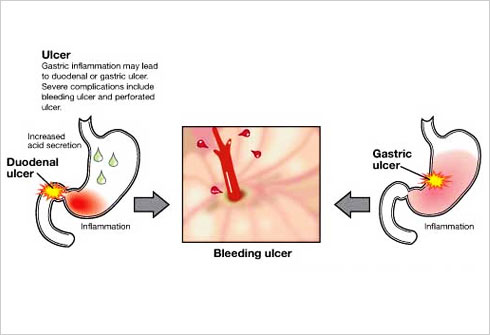
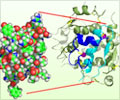
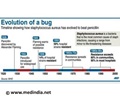
When studying the chromosomes of Tetrahymena, a unicellular ciliate organism, Elizabeth Blackburn identified a DNA sequence that was repeated several times at the ends of the chromosomes. The function of this sequence, CCCCAA, was unclear. At the same time, Jack Szostak had made the observation that a linear DNA molecule, a type of minichromosome, is rapidly degraded when introduced into yeast cells.

Both of them decided to perform an experiment.From the DNA of Tetrahymena, Blackburn isolated the CCCCAA sequence. Szostak coupled it to the minichromosomes and put them back into yeast cells. The results were striking the telomere DNA sequence protected the minichromosomes from degradation.

As telomere DNA from one organism, Tetrahymena, protected chromosomes in an entirely different one, yeast, this demonstrated the existence of a previously unrecognized fundamental mechanism.
 Angioplasty
Angioplasty
 Cardiopulmonary Resuscitation
Cardiopulmonary Resuscitation
 Heart Attack
Heart Attack
 Coronary Artery Bypass Surgery
Coronary Artery Bypass Surgery
 Mitral Valve Disease and Replacement
Mitral Valve Disease and Replacement
 Atrial Septal Defect
Atrial Septal Defect
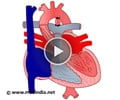 Ventricular Septal Defect
Ventricular Septal Defect
 Active Heart
Active Heart
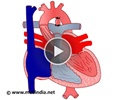 Patent Ductus Arteriosus
Patent Ductus Arteriosus
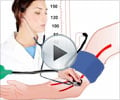 Blood Pressure
Blood Pressure
 Learn How to Count Your Pulse
Learn How to Count Your Pulse
 Angioplasty - 3D
Angioplasty - 3D
 How to know if someone is Breathing
How to know if someone is Breathing



 The Nobel Prize is an International Award governed by the Nobel Foundation in Stockholm, Sweden. It consists of a medal, personal diploma, and a cash award.
The Nobel Prize is an International Award governed by the Nobel Foundation in Stockholm, Sweden. It consists of a medal, personal diploma, and a cash award.